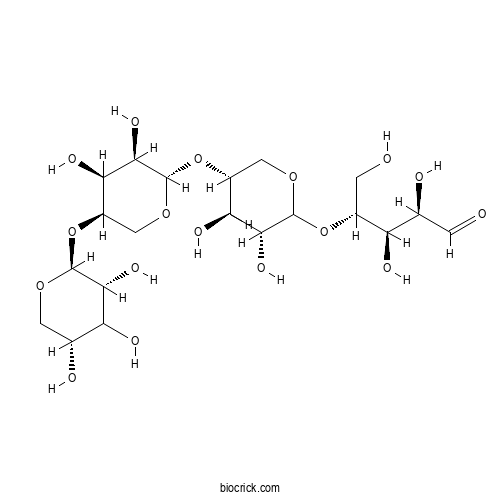
XylotetraoseCAS# 22416-58-6 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 22416-58-6 | SDF | Download SDF |
| PubChem ID | 156620289.0 | Appearance | Powder |
| Formula | C20H34O17 | M.Wt | 546.47 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2R,3R,4R)-4-[(3R,4R,5R)-5-[(2S,3R,4S,5R)-3,4-dihydroxy-5-[(2S,3R,5R)-3,4,5-trihydroxyoxan-2-yl]oxyoxan-2-yl]oxy-3,4-dihydroxyoxan-2-yl]oxy-2,3,5-trihydroxypentanal | ||
| SMILES | C1C(C(C(C(O1)OC2COC(C(C2O)O)OC3COC(C(C3O)O)OC(CO)C(C(C=O)O)O)O)O)O | ||
| Standard InChIKey | JVZHSOSUTPAVII-MIDOVEIXSA-N | ||
| Standard InChI | InChI=1S/C20H34O17/c21-1-6(23)11(25)8(2-22)35-19-16(30)13(27)10(4-33-19)37-20-17(31)14(28)9(5-34-20)36-18-15(29)12(26)7(24)3-32-18/h1,6-20,22-31H,2-5H2/t6-,7+,8+,9+,10+,11+,12?,13-,14+,15+,16+,17+,18-,19?,20-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Xylotetraose Dilution Calculator

Xylotetraose Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8299 mL | 9.1496 mL | 18.2993 mL | 36.5985 mL | 45.7482 mL |
| 5 mM | 0.366 mL | 1.8299 mL | 3.6599 mL | 7.3197 mL | 9.1496 mL |
| 10 mM | 0.183 mL | 0.915 mL | 1.8299 mL | 3.6599 mL | 4.5748 mL |
| 50 mM | 0.0366 mL | 0.183 mL | 0.366 mL | 0.732 mL | 0.915 mL |
| 100 mM | 0.0183 mL | 0.0915 mL | 0.183 mL | 0.366 mL | 0.4575 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 16-Hydroxyhexadecanoic acid
Catalog No.:BCX0812
CAS No.:506-13-8
- Monascorubrin
Catalog No.:BCX0811
CAS No.:13283-90-4
- Acetyl Dopamine Dimer I
Catalog No.:BCX0810
CAS No.:315188-82-0
- guan-fu base I
Catalog No.:BCX0809
CAS No.:110225-59-7
- Monascin
Catalog No.:BCX0808
CAS No.:21516-68-7
- Thymoquinone
Catalog No.:BCX0807
CAS No.:490-91-5
- Neoeuonymine
Catalog No.:BCX0806
CAS No.:33510-25-7
- Cortodoxone
Catalog No.:BCX0805
CAS No.:152-58-9
- Cortisone
Catalog No.:BCX0804
CAS No.:53-06-5
- Quercetin 7-O-glucuronide
Catalog No.:BCX0803
CAS No.:38934-20-2
- Coreopsin
Catalog No.:BCX0802
CAS No.:499-29-6
- Harmalan
Catalog No.:BCX0801
CAS No.:525-41-7
- Nepetalacton
Catalog No.:BCX0814
CAS No.:21651-62-7
- Kuwanon U
Catalog No.:BCX0815
CAS No.:123702-95-4
- Xylopentaose
Catalog No.:BCX0816
CAS No.:49694-20-4
- Hirudonucleodisulfide A
Catalog No.:BCX0817
CAS No.:1072789-37-7
- Xylohexaose
Catalog No.:BCX0818
CAS No.:49694-21-5
- Coreoside B
Catalog No.:BCX0819
CAS No.:1580464-83-0
- (25R)-26-O-β-D-Glucopyranosyl-22-hydroxy-5β-furost-3β,26-diol-3-O-β-D-glucopyranosyl-(1→2)-β-D-galactopyranoside
Catalog No.:BCX0820
CAS No.:897386-27-5
- Tribuloside
Catalog No.:BCX0821
CAS No.:22153-44-2
- 6''- methyl glycyrrhizinate
Catalog No.:BCX0822
CAS No.:1186016-30-7
- Picfeltarraegenin I
Catalog No.:BCX0823
CAS No.:82145-63-9
- Licorice glycoside C2
Catalog No.:BCX0824
CAS No.:202657-55-4
- Heptasaccharide
Catalog No.:BCX0825
CAS No.:121591-98-8
Leucine rich repeat-malectin receptor kinases IGP1/CORK1, IGP3 and IGP4 are required for arabidopsis immune responses triggered by beta-1,4-D-Xylo-oligosaccharides from plant cell walls.[Pubmed:38600908]
Cell Surf. 2024 Apr 4;11:100124.
Pattern-Triggered Immunity (PTI) in plants is activated upon recognition by Pattern Recognition Receptors (PRRs) of Damage- and Microbe-Associated Molecular Patterns (DAMPs and MAMPs) from plants or microorganisms, respectively. An increasing number of identified DAMPs/MAMPs are carbohydrates from plant cell walls and microbial extracellular layers, which are perceived by plant PRRs, such as LysM and Leucine Rich Repeat-Malectin (LRR-MAL) receptor kinases (RKs). LysM-RKs (e.g. CERK1, LYK4 and LYK5) are needed for recognition of fungal MAMP chitohexaose (beta-1,4-D-(GlcNAc)(6), CHI6), whereas IGP1/CORK1, IGP3 and IGP4 LRR-MAL RKs are required for perception of beta-glucans, like cellotriose (beta-1,4-D-(Glc)(3), CEL3) and mixed-linked glucans. We have explored the diversity of carbohydrates perceived by Arabidopsis thaliana seedlings by determining PTI responses upon treatment with different oligosaccharides and polysaccharides. These analyses revealed that plant oligosaccharides from xylans [beta-1,4-D-(xylose)(4) (XYL4)], glucuronoxylans and alpha-1,4-glucans, and polysaccharides from plants and seaweeds activate PTI. Cross-elicitation experiments of XYL4 with other glycans showed that the mechanism of recognition of XYL4 and the DAMP 3(3)-alpha-L-arabinofuranosyl-Xylotetraose (XA(3)XX) shares some features with that of CEL3 but differs from that of CHI6. Notably, XYL4 and XA(3)XX perception is impaired in igp1/cork1, igp3 and igp4 mutants, and almost not affected in cerk1 lyk4 lyk5 triple mutant. XYL4 perception is conserved in different plant species since XYL4 pre-treatment triggers enhanced disease resistance in tomato to Pseudomonas syringae pv tomato DC3000 and PTI responses in wheat. These results expand the number of glycans triggering plant immunity and support IGP1/CORK1, IGP3 and IGP4 relevance in Arabidopsis thaliana glycans perception and PTI activation. SIGNIFICANCE STATEMENT: The characterization of plant immune mechanisms involved in the perception of carbohydrate-based structures recognized as DAMPs/MAMPs is needed to further understand plant disease resistance modulation. We show here that IGP1/CORK1, IGP3 and IGP4 LRR-MAL RKs are required for the perception of carbohydrate-based DAMPs beta-1,4-D-(xylose)(4) (XYL4) and 3(3)-alpha-L-arabinofuranosyl-Xylotetraose (XA(3)XX), further expanding the function of these LRR-MAL RKs in plant glycan perception and immune activation.
A novel glycoside hydrolase 43-like enzyme from Clostridium boliviensis is an endo-xylanase and a candidate for xylooligosaccharide production from different xylan substrates.[Pubmed:38497645]
Appl Environ Microbiol. 2024 Apr 17;90(4):e0222323.
An uncharacterized gene encoding a glycoside hydrolase family 43-like enzyme from Clostridium boliviensis strain E-1 was identified from genomic sequence data, and the encoded enzyme, CbE1Xyn43-l, was produced in Escherichia coli. CbE1Xyn43-l (52.9 kDa) is a two-domain endo-beta-xylanase consisting of a C-terminal CBM6 and a GH43-like catalytic domain. The positions of the catalytic dyad conserved in GH43, the catalytic base (Asp74), and proton donor (Glu240) were identified in alignments including GH43-enzymes of known 3D-structure from different subfamilies. CbE1Xyn43-l is active at pH 7.0-9.0, with optimum temperature at 65 degrees C, and a more than 7 days' half-life in irreversible deactivation studies at this temperature. The enzyme hydrolyzed birchwood xylan, quinoa stalks glucuronoarabinoxylan, and wheat arabinoxylan with xylotriose and Xylotetraose as major hydrolysis products. CbE1Xyn43-l also released xylobiose from pNPX(2) with low turnover (k(cat) of 0.044 s(-1)) but was inactive on pNPX, showing that a degree of polymerization of three (DP3) was the smallest hydrolyzable substrate. Divalent ions affected the specific activity on xylan substrates, which dependent on the ion could be increased or decreased. In conclusion, CbE1Xyn43-l from C. boliviensis strain E-1 is the first characterized member of a large group of homologous hypothetical proteins annotated as GH43-like and is a thermostable endo-xylanase, producing xylooligosaccharides of high DP (xylotriose and Xylotetraose) producer. IMPORTANCE: The genome of Clostridium boliviensis strain E-1 encodes a number of hypothetical enzymes, annotated as glycoside hydrolase-like but not classified in the Carbohydrate Active Enzyme Database (CAZy). A novel thermostable GH43-like enzyme is here characterized as an endo-beta-xylanase of interest in the production of prebiotic xylooligosaccharides (XOs) from different xylan sources. CbE1Xyn43-l is a two-domain enzyme composed of a catalytic GH43-l domain and a CBM6 domain, producing xylotriose as main XO product. The enzyme has homologs in many related Clostridium strains which may indicate a similar function and be a previously unknown type of endo-xylanase in this evolutionary lineage of microorganisms.
Accelerated Lignocellulosic Molecule Adsorption Structure Determination.[Pubmed:38408381]
J Chem Theory Comput. 2024 Mar 12;20(5):2297-2312.
Here, we present a study combining Bayesian optimization structural inference with the machine learning interatomic potential Neural Equivariant Interatomic Potential (NequIP) to accelerate and enable the study of the adsorption of the conformationally flexible lignocellulosic molecules beta-d-xylose and 1,4-beta-d-Xylotetraose on a copper surface. The number of structure evaluations needed to map out the relevant potential energy surfaces are reduced by Bayesian optimization, while NequIP minimizes the time spent on each evaluation, ultimately resulting in cost-efficient and reliable sampling of large systems and configurational spaces. Although the applicability of Bayesian optimization for the conformational analysis of the more flexible Xylotetraose molecule is restricted by the sample complexity bottleneck, the latter can be effectively bypassed with external conformer search tools, such as the Conformer-Rotamer Ensemble Sampling Tool, facilitating the subsequent lower-dimensional global minimum adsorption structure determination. Finally, we demonstrate the applicability of the described approach to find adsorption structures practically equivalent to the density functional theory counterparts at a fraction of the computational cost.
A Glycosyl Hydrolase 30 Family Xylanase from the Rumen Metagenome and Its Effects on In Vitro Ruminal Fermentation of Wheat Straw.[Pubmed:38200851]
Animals (Basel). 2023 Dec 28;14(1):118.
The challenge of wheat straw as a ruminant feed is its low ruminal digestibility. This study investigated the impact of a xylanase called RuXyn, derived from the rumen metagenome of beef cattle, on the in vitro ruminal fermentation of wheat straw. RuXyn encoded 505 amino acids and was categorized within subfamily 8 of the glycosyl hydrolase 30 family. RuXyn was heterologously expressed in Escherichia coli and displayed its highest level of activity at pH 6.0 and 40 degrees C. RuXyn primarily hydrolyzed xylan, while it did not show any noticeable activity towards other substrates, including carboxymethylcellulose and Avicel. At concentrations of 5 mM, Mn(2+) and dithiothreitol significantly enhanced RuXyn's activity by 73% and 20%, respectively. RuXyn's activity was almost or completely inactivated in the presence of Cu(2+), even at low concentrations. The main hydrolysis products of corncob xylan by RuXyn were xylopentose, xylotriose, and Xylotetraose. RuXyn hydrolyzed wheat straw and rice straw more effectively than it did other agricultural by-products. A remarkable synergistic effect was observed between RuXyn and a cellulase cocktail on wheat straw hydrolysis. Supplementation with RuXyn increased dry matter digestibility; acetate, propionate, valerate, and total volatile fatty acid yields; NH(3)-N concentration, and total bacterial number during in vitro fermentation of wheat straw relative to the control. RuXyn's inactivity at 60 degrees C and 70 degrees C was remedied by mutating proline 151 to phenylalanine and aspartic acid 204 to leucine, boosting activity to 20.3% and 21.8% of the maximum activity at the respective temperatures. As an exogenous enzyme preparation, RuXyn exhibits considerable potential to improve ruminal digestion and the utilization of wheat straw in ruminants. As far as we know, this is the first study on a GH30 xylanase promoting the ruminal fermentation of agricultural straws. The findings demonstrate that the utilization of RuXyn can significantly enhance the ruminal digestibility of wheat straw by approximately 10 percentage points. This outcome signifies the emergence of a novel and highly efficient enzyme preparation that holds promise for the effective utilization of wheat straw, a by-product of crop production, in ruminants.
Efficient production of xylooligosaccharides from Camellia oleifera shells pretreated by pyruvic acid at lower temperature.[Pubmed:38199559]
Int J Biol Macromol. 2024 Feb;259(Pt 2):129262.
XOS production from lignocellulose using organic carboxylic acids and alkyd acids has been widely reported. However, it still faces harsh challenges such as high energy consumption, high cost, and low purity. Pyruvic acid (PYA), a carbonyl acid with carbonyl and carboxyl groups, was used to produce XOS due to its stronger catalytic activity. In this work, XOS was efficiently prepared from COS in an autoclave under the condition of 0.21 M PYA-121 degrees C-35 min. The total yield of XOS reached 68.72 % without producing any toxic by-products, including furfural (FF) and 5-hydroxymethylfurfural (5-HMF). The yield of xylobiose (X(2)), xylotriose (X(3)), Xylotetraose (X(4)), and xylopentaose (X(5)) were 20.58 %, 12.47 %, 15.74 %, and 10.05 %, respectively. Meanwhile, 89.05 % of lignin was retained in the solid residue, which provides a crucial functional group for synthesizing layered carbon materials (SRG-a). It achieves excellent electromagnetic shielding (EMS) performance through graphitization, reaching -30 dB at a thickness of 2.0 mm. The use of a PYA catalyst in the production of XOS has proven to be an efficient method due to lower temperature, lower acid consumption, and straightforward operation.
Biochemical unravelling of the endoxylanase activity in a bifunctional GH39 enzyme cloned and expressed from thermophilic Geobacillus sp. WSUCF1.[Pubmed:38072346]
Int J Biol Macromol. 2024 Feb;257(Pt 2):128679.
The glycoside hydrolase family 39 (GH39) proteins are renowned for their extremophilic and multifunctional enzymatic properties, yet the molecular mechanisms underpinning these unique characteristics continue to be an active subject of research. In this study, we introduce WsuXyn, a GH39 protein with a molecular weight of 58 kDa, originating from the thermophilic Geobacillus sp. WSUCF1. Previously reported for its exceptional thermostable beta-xylosidase activity, WsuXyn has recently demonstrated a significant endoxylanase activity (3752 U.mg(-1)) against beechwood xylan, indicating towards its bifunctional nature. Physicochemical characterization revealed that WsuXyn exhibits optimal endoxylanase activity at 70 degrees C and pH 7.0. Thermal stability assessments revealed that the enzyme is resilient to elevated temperatures, with a half-life of 168 h. Key kinetic parameters highlight the exceptional catalytic efficiency and strong affinity of the protein for xylan substrate. Moreover, WsuXyn-mediated hydrolysis of beechwood xylan has achieved 77 % xylan conversion, with xylose as the primary product. Structural analysis, amalgamated with docking simulations, has revealed strong binding forces between Xylotetraose and the protein, with key amino acid residues, including Glu278, Tyr230, Glu160, Gly202, Cys201, Glu324, and Tyr283, playing pivotal roles in these interactions. Therefore, WsuXyn holds a strong promise for biodegradation and value-added product generation through lignocellulosic biomass conversion.
Induction of plant disease resistance by mixed oligosaccharide elicitors prepared from plant cell wall and crustacean shells.[Pubmed:37882264]
Physiol Plant. 2023 Sep-Oct;175(5):e14052.
Basal plant immune responses are activated by the recognition of conserved microbe-associated molecular patterns (MAMPs), or breakdown molecules released from the plants after damage by pathogen penetration, so-called damage-associated molecular patterns (DAMPs). While chitin-oligosaccharide (CHOS), a primary component of fungal cell walls, is most known as MAMP, plant cell wall-derived oligosaccharides, cello-oligosaccharides (COS) from cellulose, and xylo-oligosaccharide (XOS) from hemicellulose are representative DAMPs. In this study, elicitor activities of COS prepared from cotton linters, XOS prepared from corn cobs, and chitin-oligosaccharide (CHOS) from crustacean shells were comparatively investigated. In Arabidopsis, COS, XOS, or CHOS treatment triggered typical defense responses such as reactive oxygen species (ROS) production, phosphorylation of MAP kinases, callose deposition, and activation of the defense-related transcription factor WRKY33 promoter. When COS, XOS, and CHOS were used at concentrations with similar activity in inducing ROS production and callose depositions, CHOS was particularly potent in activating the MAPK kinases and WRKY33 promoters. Among the COS and XOS with different degrees of polymerization, cellotriose and Xylotetraose showed the highest activity for the activation of WRKY33 promoter. Gene ontology enrichment analysis of RNAseq data revealed that simultaneous treatment of COS, XOS, and CHOS (oligo-mix) effectively activates plant disease resistance. In practice, treatment with the oligo-mix enhanced the resistance of tomato to powdery mildew, but plant growth was not inhibited but rather tended to be promoted, providing evidence that treatment with the oligo-mix has beneficial effects on improving disease resistance in plants, making them a promising class of compounds for practical application.
Characterization of a GH10 extremely thermophilic xylanase from the metagenome of hot spring for prebiotic production.[Pubmed:37749183]
Sci Rep. 2023 Sep 25;13(1):16053.
A xylanase gene (named xyngmqa) was identified from the metagenomic data of the Gumingquan hot spring (92.5 degrees C, pH 9.2) in Tengchong City, Yunnan Province, southwest China. It showed the highest amino acid sequence identity (82.70%) to endo-1,4-beta-xylanase from Thermotoga caldifontis. A constitutive expression plasmid (denominated pSHY211) and double-layer plate (DLP) method were constructed for cloning, expression, and identification of the XynGMQA gene. The XynGMQA gene was synthesized and successfully expressed in Escherichia coli DH5alpha. XynGMQA exhibited optimal activity at 90 degrees C and pH 4.6, being thermostable by maintaining 100% of its activity after 2 h incubated at 80 degrees C. Interestingly, its enzyme activity was enhanced by high temperatures (70 and 80 degrees C) and low pH (3.0-6.0). About 150% enzyme activity was detected after incubation at 70 degrees C for 20 to 60 min or 80 degrees C for 10 to 40 min, and more than 140% enzyme activity after incubation at pH 3.0 to 6.0 for 12 h. Hydrolytic products of beechwood xylan with XynGMQA were xylooligosaccharides, including xylobiose (X2), xylotriose (X3), and Xylotetraose (X4). These properties suggest that XynGMQA as an extremely thermophilic xylanase, may be exploited for biofuel and prebiotic production from lignocellulosic biomass.
Xylanopectinolytic enzymes by marine actinomycetes from sediments of Sarena Kecil, North Sulawesi: high potential to produce galacturonic acid and xylooligosaccharides from raw biomass.[Pubmed:36920661]
J Genet Eng Biotechnol. 2023 Mar 15;21(1):31.
BACKGROUND: Actinomycetes isolated from marine habitats are known to have the potential for novel enzymes that are beneficial in the industry. In-depth knowledge is necessary given the variety of this bacterial group in Indonesia and the lack of published research. Actinomycetes isolates (BLH 5-14) obtained from marine sediments of Sarena Kecil, Bitung, North Sulawesi, Indonesia, showed an ability to produce pectinase and xylanase that have equal or even higher potential for pectic-oligosaccharides (POS) and xylooligosaccharides (XOS) production from raw biomass than from commercial substrates. This study's objective was to characterize both enzymes to learn more for future research and development. RESULTS: Pectinase had the highest activity on the 6(th) day (1.44+/-0.08 U/mL) at the optimum pH of 8.0 and optimum temperature of 50 degrees C. Xylanase had the maximum activity on the 6(th) day (4.33+/-0.03 U/mL) at optimum pH 6.0 and optimum temperature 60 degrees C. Hydrolysis and thin layer chromatography also showed that pectinase was able to produce monosaccharides such as galacturonic acid (P1), and xylanase was able to yield oligosaccharides such as xylotriose (X3), Xylotetraose (X4), and xylopentaose (X5). BLH 5-14 identified as the genus Streptomyces based on the 16S rDNA sequences and the closely related species Streptomyces tendae (99,78%). CONCLUSIONS: In the eco-friendly paper bleaching industry, Streptomyces tendae has demonstrated the potential to create enzymes with properties that can be active in a wide range of pH levels. The oligosaccharides have the potential as prebiotics or dietary supplements with anti-cancer properties. Further research is needed to optimize the production, purification, and development of the application of pectinase and xylanase enzymes produced by Actinomycetes isolates.
Engineering mesophilic GH11 xylanase from Cellulomonas flavigena by rational design of N-terminus substitution.[Pubmed:36406235]
Front Bioeng Biotechnol. 2022 Nov 3;10:1044291.
Xylanase, a glycoside hydrolase, is widely used in the food, papermaking, and textile industries; however, most xylanases are inactive at high temperatures. In this study, a xylanase gene, CFXyl3, was cloned from Cellulomonas flavigena and expressed in Escherichia coli BL21 (DE3). To improve the thermostability of xylanase, four hybrid xylanases with enhanced thermostability (designated EcsXyl1-4) were engineered from CFXyl3, guided by primary and 3D structure analyses. The optimal temperature of CFXyl3 was improved by replacing its N-terminus with the corresponding area of SyXyn11P, a xylanase that belongs to the hyperthermostable GH11 family. The optimal temperatures of the hybrid xylanases EcsXyl1-4 were 60, 60, 65, and 85 degrees C, respectively. The optimal temperature of EcsXyl4 was 30 C higher than that of CFXyl3 (55 degrees C) and its melting temperature was 34.5 degrees C higher than that of CFXyl3. After the hydrolysis of beechwood xylan, the main hydrolysates were Xylotetraose, xylotriose, and xylobiose; thus, these hybrid xylanases could be applied to prebiotic xylooligosaccharide manufacturing.
Characterization of a xylanase belonging to the glycoside hydrolase family 5 subfamily 35 from Paenibacillus sp. H2C.[Pubmed:36352459]
Biosci Biotechnol Biochem. 2022 Dec 21;87(1):54-62.
Corn xylan is resistant to enzymatic hydrolysis due to its complex structure. We characterized PsXyn5A, an enzyme highly active for corn xylan, isolated from Paenibacillus sp. H2C. PsXyn5A is a modular xylanase with a catalytic domain belonging to the glycoside hydrolase family 5 subfamily 35 (GH5_35) and a carbohydrate-binding module family 13 (CBM13) domain. The substrate recognition mechanism of GH5_35 xylanase has not been reported. Analysis of the hydrolysate from rye arabinoxylan (RAX) has shown that the GH5_35 catalytic domain of PsXyn5A recognizes an arabinofuranosyl (Araf) side residue and cleaves the reducing terminal side of Araf-linked xylopyranose. This cleavage specificity is the same as reported for the GH5_34 xylanase from Hungateiclostridium thermocellum (HtXyl5A). Unlike HtXyl5A, PsXyn5A produced Araf-xylopyranose from RAX and did not hydrolyze 33-alpha-l-Araf-Xylotetraose. Deletion of the CBM13 domain significantly decreased the activity toward insoluble corn xylan, indicating that CBM13 plays an essential role in hydrolyzing corn xylan.
Characteristics of recombinant xylanase from camel rumen metagenome and its effects on wheat bran hydrolysis.[Pubmed:36027987]
Int J Biol Macromol. 2022 Nov 1;220:1309-1317.
In the present study, we explored the effects of a novel xylanase from camel rumen metagenome (CrXyn) on wheat bran hydrolysis. CrXyn was heterologously expressed in Escherichia coli and showed maximum activity at 40 degrees C and pH 7.0. Furthermore, CrXyn exhibited preferential hydrolysis of xylan, but no obvious activity toward other substrates, including carboxymethylcellulose and Avicel. Using wheat straw xylan as a substrate, the Km and Vmax values for CrXyn were 5.98 g/L and 179.9 mumol xylose/min/mg protein, respectively. Mn(2+) was a strong accelerator and significantly enhanced CrXyn activity. However, CrXyn activity was inhibited (~50 %) by 1 mM and 5 mM ethylenediaminetetraacetic acid (EDTA) and completely inactivated by 5 mM Cu(2+). CrXyn tolerated 5 mM sodium dodecyl sulphate (SDS) and 15 % methanol, ethanol, and dimethyl sulfoxide (DMSO), with >50 % residual activity. CrXyn effectively hydrolyzed wheat bran, with xylobiose and Xylotetraose accounting for 79.1 % of total sugars produced. A remarkable synergistic effect was found between CrXyn and protease, leading to an obvious increase in amino acids released from wheat bran compared with the control. CrXyn also enhanced the in vitro hydrolysis of wheat bran. Thus, CrXyn exhibits great potential as a feed additive to improve the utilization of wheat bran in monogastric animal production.
Chemical and Enzymatic Synthesis of Biobased Xylo-Oligosaccharides and Fermentable Sugars from Wheat Straw for Food Applications.[Pubmed:35406211]
Polymers (Basel). 2022 Mar 25;14(7):1336.
Xylo-oligosaccharides are sugar oligomers with 2~7 xylose units considered non-digestible fibers that can be produced from biodegradable and low-cost biomass like wheat straw. An integrated approach consisting of hydrothermal pretreatment, alkaline treatment, enzymatic treatment and the combinations thereof was applied to overcome the recalcitrance structure of the wheat straw and allow selective fractioning into fermentable sugars and xylo-oligosaccharides. The hydrolysates and processed solids were chemically characterized by High-performance liquid chromatography and Ion chromatography, and the results were expressed as function of the severity factor and statistically interpreted. The concentration of fermentable sugars (glucose, xylose, arabinose) was the highest after the combination of alkaline and enzymatic treatment with xylanase (18 g/L sugars), while xylo-oligosaccharides (xylotriose and Xylotetraose) were released in lower amounts (1.33 g/L) after the same treatment. Refining experiments were carried out to obtain a purified fraction by using anion and cation exchange chromatography. The polymer adsorber resin MN-502 showed efficient removal of salts, phenols and furan derivatives. However, the xylo-oligosaccharides yields were also slightly reduced. Although still requiring further optimization of the treatments to obtain higher purified oligomer yields, the results provide information on the production of xylo-oligosaccharides and fermentable sugars from wheat straw for potential use in food applications.
Biochemical Basis of Xylooligosaccharide Utilisation by Gut Bacteria.[Pubmed:35328413]
Int J Mol Sci. 2022 Mar 10;23(6):2992.
Xylan is one of the major structural components of the plant cell wall. Xylan present in the human diet reaches the large intestine undigested and becomes a substrate to species of the gut microbiota. Here, we characterised the capacity of Limosilactobacillus reuteri and Blautia producta strains to utilise xylan derivatives. We showed that L. reuteri ATCC 53608 and B. producta ATCC 27340 produced beta-D-xylosidases, enabling growth on xylooligosaccharide (XOS). The recombinant enzymes were highly active on artificial (p-nitrophenyl beta-D-xylopyranoside) and natural (xylobiose, xylotriose, and Xylotetraose) substrates, and showed transxylosylation activity and tolerance to xylose inhibition. The enzymes belong to glycoside hydrolase family 120 with Asp as nucleophile and Glu as proton donor, as shown by homology modelling and confirmed by site-directed mutagenesis. In silico analysis revealed that these enzymes were part of a gene cluster in L. reuteri but not in Blautia strains, and quantitative proteomics identified other enzymes and transporters involved in B. producta XOS utilisation. Based on these findings, we proposed a model for an XOS metabolism pathway in L. reuteri and B. producta strains. Together with phylogenetic analyses, the data also revealed the extended xylanolytic potential of the gut microbiota.
Application of a recombinant GH10 endoxylanase from Thermoascus aurantiacus for xylooligosaccharide production from sugarcane bagasse and probiotic bacterial growth.[Pubmed:35151712]
J Biotechnol. 2022 Mar 10;347:1-8.
Xylooligosaccharides (XOs) are a promising class of prebiotics capable of selectively stimulating the growth of the beneficial intestinal microbiota against intestinal pathogens. They can be obtained from xylan present in residual lignocellulosic material from agriculture. Thus, in this study we produced XOs by extracting xylan from sugarcane bagasse and hydrolyzing it using the GH10 xylanase from Thermoascus aurantiacus expressed by Pichia pastoris. An alkaline method to extract xylan is described, which resulted in 83.40% of xylan recovery and low amounts of cellulose and lignin. The enzymatic hydrolysate exhibited a mixture of XOs containing mainly xylobiose, xylotriose and Xylotetraose. These oligosaccharides stimulated the growth of Lactobacillus casei, L. rhamnosus, L. fermentum and L. bulgaricus strains, which were able to produce organic acids, especially acetic acid. These findings demonstrate the possibility to redirect crop by-products to produce XOs and their use as a supplement to stimulate the growth of probiotic strains.


