Clozapine N-oxide (CNO)Metabolite of clozapine, used in chemogenetics. CAS# 34233-69-7 |
- Prasugrel
Catalog No.:BCC1089
CAS No.:150322-43-3
Quality Control & MSDS
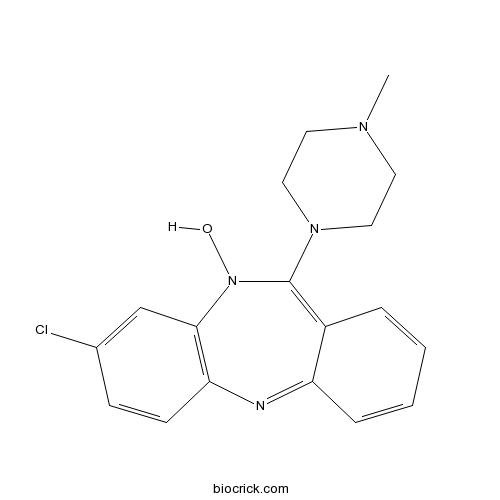
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 34233-69-7 | SDF | Download SDF |
| PubChem ID | 36727 | Appearance | Powder |
| Formula | C18H19ClN4O | M.Wt | 342.82 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 20 mM in DMSO and to 20 mM in water | ||
| Chemical Name | 3-chloro-5-hydroxy-6-(4-methylpiperazin-1-yl)benzo[b][1,4]benzodiazepine | ||
| SMILES | CN1CCN(CC1)C2=C3C=CC=CC3=NC4=C(N2O)C=C(C=C4)Cl | ||
| Standard InChIKey | YWSTWDVPYHSYLN-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C18H19ClN4O/c1-21-8-10-22(11-9-21)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)23(18)24/h2-7,12,24H,8-11H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Metabolite of clozapine. Activates human muscarinic designer receptors (DREADDs). Silences hippocampal neurons expressing hM4 DREADDs in vitro. Suppresses food intake in hyperphagic mice with hM3Dq-expressing, LMO4-deficient, paraventricular hypothalamic neurons. |

Clozapine N-oxide (CNO) Dilution Calculator

Clozapine N-oxide (CNO) Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.917 mL | 14.5849 mL | 29.1698 mL | 58.3397 mL | 72.9246 mL |
| 5 mM | 0.5834 mL | 2.917 mL | 5.834 mL | 11.6679 mL | 14.5849 mL |
| 10 mM | 0.2917 mL | 1.4585 mL | 2.917 mL | 5.834 mL | 7.2925 mL |
| 50 mM | 0.0583 mL | 0.2917 mL | 0.5834 mL | 1.1668 mL | 1.4585 mL |
| 100 mM | 0.0292 mL | 0.1458 mL | 0.2917 mL | 0.5834 mL | 0.7292 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Clozapine-N-oxide (CNO) is a metabolite of clozapine, which reduces the density of 5-HT2 receptor in rat primary cortical cells. [1] Clozapine-N-oxide (CNO) also activates an evolved M3-muscarinic receptor and then can be used in chemogenetics. [2]
Clozapine is a known atypical antipsychotic medication used in the treatment of schizophrenia. The two major metabolites of clozapine are norclozapine and clozapine-N-oxide (CNO) [3]. Clozapine is also formed from its N-oxide metabolite. It means that a reversible metabolic pathway exists for clozapine and clozapine-N-oxide (CNO) [4]. In rat primary cortical cells and SHS5Y5 neuroblastoma cells, a significant decrease was found for 5-HT2 receptor density with 200 ng/ml clozapine-N-oxide (CNO) [1]. About the method of identification, reversed-phase HPLC has been reported to detect clozapine and its two major metabolites in human plasma[5].
References:
1. P. Heiser, E. Schulte, C. Hausmann, R. Becker, H. Remschmidt, J. C. Krieg and H. Vedder, Prog Neuropsychopharmacol Biol Psychiatry 2004, 28, 297-302.
2. Armbruster BN1, Li X, Pausch MH, et al. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc Natl Acad Sci U S A. 2007 Mar 20;104(12):5163-8.
3. S. A. Volpicelli, F. Centorrino, P. R. Puopolo, J. Kando, F. R. Frankenburg, R. J. Baldessarini and J. G. Flood, Clin Chem 1993, 39, 1656-1659.
4. M. W. Jann, Y. W. Lam and W. H. Chang, Arch Int Pharmacodyn Ther 1994, 328, 243-250.
5. A. Avenoso, G. Facciola, G. M. Campo, A. Fazio and E. Spina, J Chromatogr B Biomed Sci Appl 1998, 714, 299-308.
- Tropine acetate
Catalog No.:BCN1922
CAS No.:3423-27-6
- 3-Acetoxytropane
Catalog No.:BCN1933
CAS No.:3423-26-5
- Echinatin
Catalog No.:BCN6277
CAS No.:34221-41-5
- Tovopyrifolin C
Catalog No.:BCN6888
CAS No.:34211-53-5
- Trachelogenin
Catalog No.:BCN2739
CAS No.:34209-69-3
- 7-beta-Hydroxylathyrol
Catalog No.:BCN3413
CAS No.:34208-98-5
- Dihydroflavokawin B
Catalog No.:BCN5272
CAS No.:3791-76-2
- 3-Methoxyfuran
Catalog No.:BCN5271
CAS No.:3420-57-3
- Lys-Bradykinin
Catalog No.:BCC5993
CAS No.:342-10-9
- Actinidic acid
Catalog No.:BCN5270
CAS No.:341971-45-7
- Propafenone HCl
Catalog No.:BCC5079
CAS No.:34183-22-7
- Pterosin B
Catalog No.:BCN7100
CAS No.:34175-96-7
- Telbivudine
Catalog No.:BCC3862
CAS No.:3424-98-4
- Saralasin
Catalog No.:BCC5714
CAS No.:34273-10-4
- Angiotensin 1/2 (1-9)
Catalog No.:BCC1005
CAS No.:34273-12-6
- Gardneramine
Catalog No.:BCN5273
CAS No.:34274-91-4
- Urotensin II-related peptide
Catalog No.:BCC5884
CAS No.:342878-90-4
- SDM25N hydrochloride
Catalog No.:BCC7054
CAS No.:342884-71-3
- Chikusetsusaponin V methyl ester
Catalog No.:BCN3472
CAS No.:34291-22-0
- 2,3-Dihydrohinokiflavone
Catalog No.:BCN6680
CAS No.:34292-87-0
- 5,6-Dimethoxy-2-isopropenylbenzofuran
Catalog No.:BCN7195
CAS No.:34293-09-9
- Harmine hydrochloride
Catalog No.:BCN2485
CAS No.:343-27-1
- 2,16-Kauranediol
Catalog No.:BCN5274
CAS No.:34302-37-9
- Boc-Abu-OH.DCHA
Catalog No.:BCC3200
CAS No.:27494-48-0
Chemogenetic silencing of GABAergic dorsal horn interneurons induces morphine-resistant spontaneous nocifensive behaviours.[Pubmed:28680103]
Sci Rep. 2017 Jul 5;7(1):4739.
Inhibitory interneurons in the spinal dorsal horn (SDH) are crucial for processing somatosensory information originating in the periphery. However, the effects of the acute and selective inactivation of GABAergic SDH interneurons on pain processing are not fully understood. In this study, we used designer receptors exclusively activated by designer drugs (DREADD) technology and vesicular GABA transporter-Cre (Vgat-Cre) mice to selectively express a modified human muscarinic Gi protein-coupled receptor (hM4Di) in Vgat-Cre (+) GABAergic SDH interneurons in the fourth lumbar segment. We found that clozapine-N-oxide (CNO) treatment rapidly hyperpolarized these neurons and induced spontaneous nocifensive behaviours in these mice. In Vgat-Cre (neg) lamina II neurons, CNO produced facilitation of A fibre-mediated polysynaptic excitatory responses, an effect that required N-methyl-D-aspartate (NMDA) receptor activation. The CNO-induced nocifensive behaviours were also reduced by NMDA receptor antagonism. Moreover, these nocifensive behaviours were suppressed by pregabalin but resistant to morphine. Our findings indicate that Vgat-Cre (+) SDH interneurons play an important role in morphine-resistant nocifensive behaviours and suggest that this approach may provide a useful model for understanding the mechanisms of opioid-resistant pain signalling and for developing novel analgesics.
Catecholaminergic A1/C1 neurons contribute to the maintenance of upper airway muscle tone but may not participate in NREM sleep-related depression of these muscles.[Pubmed:28711601]
Respir Physiol Neurobiol. 2017 Oct;244:41-50.
Neural mechanisms of obstructive sleep apnea, a common sleep-related breathing disorder, are incompletely understood. Hypoglossal motoneurons, which provide tonic and inspiratory activation of genioglossus (GG) muscle (a major upper airway dilator), receive catecholaminergic input from medullary A1/C1 neurons. We aimed to determine the contribution of A1/C1 neurons in control of GG muscle during sleep and wakefulness. To do so, we placed injections of a viral vector into DBH-cre mice to selectively express the hMD4i inhibitory chemoreceptors in A1/C1 neurons. Administration of the hM4Di ligand, clozapine-N-oxide (CNO), in these mice decreased GG muscle activity during NREM sleep (F1,1,3=17.1, p<0.05); a similar non-significant decrease was observed during wakefulness. CNO administration had no effect on neck muscle activity, respiratory parameters or state durations. In addition, CNO-induced inhibition of A1/C1 neurons did not alter the magnitude of the naturally occurring depression of GG activity during transitions from wakefulness to NREM sleep. These findings suggest that A1/C1 neurons have a net excitatory effect on GG activity that is most likely mediated by hypoglossal motoneurons. However, the activity of A1/C1 neurons does not appear to contribute to NREM sleep-related inhibition of GG muscle activity, suggesting that A1/C1 neurons regulate upper airway patency in a state-independent manner.
Tg(Th-Cre)FI172Gsat (Th-Cre) defines neurons that are required for full hypercapnic and hypoxic reflexes.[Pubmed:28684394]
Biol Open. 2017 Aug 15;6(8):1200-1208.
The catecholaminergic (CA) system has been implicated in many facets of breathing control and offers an important target to better comprehend the underlying etiologies of both developmental and adult respiratory pathophysiologies. Here, we used a noninvasive DREADD-based pharmacogenetic approach to acutely perturb Tg(Th-Cre)FI172Gsat (Th-Cre)-defined neurons in awake and unrestrained mice in an attempt to characterize CA function in breathing. We report that clozapine-N-oxide (CNO)-DREADD-mediated inhibition of Th-Cre-defined neurons results in blunted ventilatory responses under respiratory challenge. Under a hypercapnic challenge (5% CO2/21% O2/74% N2), perturbation of Th-Cre neurons results in reduced fR, [Formula: see text] and [Formula: see text] Under a hypoxic challenge (10% O2/90% N2), we saw reduced fR, [Formula: see text] and [Formula: see text], in addition to instability in both interbreath interval and tidal volume, resulting in a Cheyne-Stokes-like respiratory pattern. These findings demonstrate the necessity of Th-Cre-defined neurons for the hypercapnic and hypoxic ventilatory responses and breathing stability during hypoxia. However, given the expanded non-CA expression domains of the Tg(Th-Cre)FI172Gsat mouse line found in the brainstem, full phenotypic effect cannot be assigned solely to CA neurons. Nonetheless, this work identifies a key respiratory population that may lead to further insights into the circuitry that maintains respiratory stability in the face of homeostatic challenges.
DREADD-induced silencing of the medial amygdala reduces the preference for male pheromones and the expression of lordosis in estrous female mice.[Pubmed:28677202]
Eur J Neurosci. 2017 Aug;46(4):2035-2046.
Sexually naive estrous female mice seek out male urinary pheromones; however, they initially display little receptive (lordosis) behavior in response to male mounts. Vomeronasal-accessory olfactory bulb inputs to the medial amygdala (Me) regulate courtship in female rodents. We used a reversible inhibitory chemogenetic technique (Designer Receptors Exclusively Activated by Designer Drugs; DREADDs) to assess the contribution of Me signaling to females' preference for male pheromones and improvement in receptivity normally seen with repeated testing. Sexually naive females received bilateral Me injections of an adeno-associated virus carrying an inhibitory DREADD. Females were later ovariectomized, treated with ovarian hormones, and given behavioral tests following intraperitoneal injections of saline or clozapine-N-oxide (CNO; which hyperpolarizes infected Me neurons). CNO attenuated females' preference to investigate male vs. female urinary odors. Repeated CNO treatment also slowed the increase in lordosis otherwise seen in females given saline. However, when saline was given to females previously treated with CNO, their lordosis quotients were as high as other females repeatedly given saline. No disruptive behavioral effects of CNO were seen in estrous females lacking DREADD infections of the Me. Finally, CNO attenuated the ability of male pheromones to stimulate Fos expression in the Me of DREADD-infected mice but not in non-infected females. Our results affirm the importance of Me signaling in females' chemosensory preferences and in the acute expression of lordosis. However, they provide no indication that Me signaling is required for the increase in receptivity normally seen after repeated hormone priming and testing with a male.
Chemogenetics revealed: DREADD occupancy and activation via converted clozapine.[Pubmed:28774929]
Science. 2017 Aug 4;357(6350):503-507.
The chemogenetic technology DREADD (designer receptors exclusively activated by designer drugs) is widely used for remote manipulation of neuronal activity in freely moving animals. DREADD technology posits the use of "designer receptors," which are exclusively activated by the "designer drug" Clozapine N-oxide (CNO). Nevertheless, the in vivo mechanism of action of CNO at DREADDs has never been confirmed. CNO does not enter the brain after systemic drug injections and shows low affinity for DREADDs. Clozapine, to which CNO rapidly converts in vivo, shows high DREADD affinity and potency. Upon systemic CNO injections, converted clozapine readily enters the brain and occupies central nervous system-expressed DREADDs, whereas systemic subthreshold clozapine injections induce preferential DREADD-mediated behaviors.
LMO4 is essential for paraventricular hypothalamic neuronal activity and calcium channel expression to prevent hyperphagia.[Pubmed:24381275]
J Neurosci. 2014 Jan 1;34(1):140-8.
The dramatic increase in the prevalence of obesity reflects a lack of progress in combating one of the most serious health problems of this century. Recent studies have improved our understanding of the appetitive network by focusing on the paraventricular hypothalamus (PVH), a key region responsible for the homeostatic balance of food intake. Here we show that mice with PVH-specific ablation of LIM domain only 4 (Lmo4) become rapidly obese when fed regular chow due to hyperphagia rather than to reduced energy expenditure. Brain slice recording of LMO4-deficient PVH neurons showed reduced basal cellular excitability together with reduced voltage-activated Ca(2+) currents. Real-time PCR quantification revealed that LMO4 regulates the expression of Ca(2+) channels (Cacna1h, Cacna1e) that underlie neuronal excitability. By increasing neuronal activity using designer receptors exclusively activated by designer drugs technology, we could suppress food intake of PVH-specific LMO4-deficient mice. Together, these results demonstrate that reduced neural activity in LMO4-deficient PVH neurons accounts for hyperphagia. Thus, maintaining PVH activity is important to prevent hyperphagia-induced obesity.
Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand.[Pubmed:17360345]
Proc Natl Acad Sci U S A. 2007 Mar 20;104(12):5163-8.
We evolved muscarinic receptors in yeast to generate a family of G protein-coupled receptors (GPCRs) that are activated solely by a pharmacologically inert drug-like and bioavailable compound (clozapine-N-oxide). Subsequent screening in human cell lines facilitated the creation of a family of muscarinic acetylcholine GPCRs suitable for in vitro and in situ studies. We subsequently created lines of telomerase-immortalized human pulmonary artery smooth muscle cells stably expressing all five family members and found that each one faithfully recapitulated the signaling phenotype of the parent receptor. We also expressed a G(i)-coupled designer receptor in hippocampal neurons (hM(4)D) and demonstrated its ability to induce membrane hyperpolarization and neuronal silencing. We have thus devised a facile approach for designing families of GPCRs with engineered ligand specificities. Such reverse-engineered GPCRs will prove to be powerful tools for selectively modulating signal-transduction pathways in vitro and in vivo.
The involvement of CYP1A2 and CYP3A4 in the metabolism of clozapine.[Pubmed:9384460]
Br J Clin Pharmacol. 1997 Nov;44(5):439-46.
AIMS: Clozapine (CLZ), an atypical neuroleptic with a high risk of causing agranulocytosis, is metabolized in the liver to desmethylclozapine (DCLZ) and clozapine N-oxide (CLZ-NO). This study investigated the involvement of different CYP isoforms in the formation of these two metabolites. METHODS: Human liver microsomal incubations, chemical inhibitors, specific antibodies, and different cytochrome P450 expression systems were used. RESULTS: Km and Vmax values determined in human liver microsomes were lower for the demethylation (61 +/- 21 microM, 159 +/- 42 pmol min(-1) mg protein(-1) mean +/- s.d.; n = 4), than for the N-oxidation of CLZ (308 +/- 1.5 microM, 456 +/- 167 pmol min(-1) mg protein(-1); n = 3). Formation of DCLZ was inhibited by fluvoxamine (53 +/- 28% at 10 microM), triacetyloleandomycin (33 +/- 15% at 10 microM), and ketoconazole (51 +/- 28% at 2 microM) and by antibodies against CYP1A2 and CYP3A4. CLZ-NO formation was inhibited by triacetyloleandomycin (34 +/- 16% at 10 microM) and ketoconazole (51 +/- 13% at 2 microM), and by antibodies against CYP3A4. There was a significant correlation between CYP3A content and DCLZ formation in microsomes from 15 human livers (r=0.67; P=0.04). A high but not significant correlation coefficient was found for CYP3A content and CLZ-NO formation (r=0.59; P=0.09). Using expression systems it was shown that CYP1A2 and CYP3A4 formed DCLZ and CLZ-NO. Km and Vmax values were lower in the CYP1A2 expression system compared to CYP3A4 for both metabolic reactions. CONCLUSIONS: It is concluded that CYP1A2 and CYP3A4 are involved in the demethylation of CLZ and CYP3A4 in the N-oxidation of CLZ. Close monitoring of CLZ plasma levels is recommended in patients treated at the same time with other drugs affecting these two enzymes.


