Cyanopindolol hemifumarateCAS# 69906-86-1 |
- SGI-1776 free base
Catalog No.:BCC2232
CAS No.:1025065-69-3
- LKB1 (AAK1 dual inhibitor)
Catalog No.:BCC1705
CAS No.:1093222-27-5
- CX-6258
Catalog No.:BCC1504
CAS No.:1202916-90-2
- AZD1208
Catalog No.:BCC2079
CAS No.:1204144-28-4
- SMI-4a
Catalog No.:BCC2233
CAS No.:438190-29-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 69906-86-1 | SDF | Download SDF |
| PubChem ID | 45073413 | Appearance | Powder |
| Formula | C36H46N6O8 | M.Wt | 690.8 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in DMSO | ||
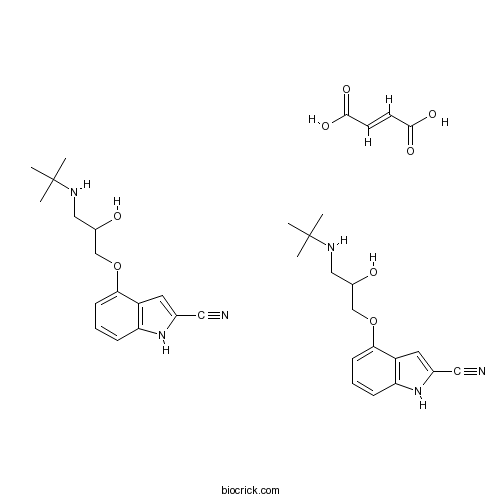
| Chemical Name | (E)-but-2-enedioic acid;4-[3-(tert-butylamino)-2-hydroxypropoxy]-1H-indole-2-carbonitrile | ||
| SMILES | CC(C)(C)NCC(COC1=CC=CC2=C1C=C(N2)C#N)O.CC(C)(C)NCC(COC1=CC=CC2=C1C=C(N2)C#N)O.C(=CC(=O)O)C(=O)O | ||
| Standard InChIKey | ZSBITJKBOWVCCI-WXXKFALUSA-N | ||
| Standard InChI | InChI=1S/2C16H21N3O2.C4H4O4/c2*1-16(2,3)18-9-12(20)10-21-15-6-4-5-14-13(15)7-11(8-17)19-14;5-3(6)1-2-4(7)8/h2*4-7,12,18-20H,9-10H2,1-3H3;1-2H,(H,5,6)(H,7,8)/b;;2-1+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 5-HT1A/1B antagonist with roughly equal affinity at each receptor; also a β-adrenoceptor antagonist. |

Cyanopindolol hemifumarate Dilution Calculator

Cyanopindolol hemifumarate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.4476 mL | 7.238 mL | 14.476 mL | 28.9519 mL | 36.1899 mL |
| 5 mM | 0.2895 mL | 1.4476 mL | 2.8952 mL | 5.7904 mL | 7.238 mL |
| 10 mM | 0.1448 mL | 0.7238 mL | 1.4476 mL | 2.8952 mL | 3.619 mL |
| 50 mM | 0.029 mL | 0.1448 mL | 0.2895 mL | 0.579 mL | 0.7238 mL |
| 100 mM | 0.0145 mL | 0.0724 mL | 0.1448 mL | 0.2895 mL | 0.3619 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Cinalbicol
Catalog No.:BCN7464
CAS No.:69904-85-4
- Immethridine dihydrobromide
Catalog No.:BCC7328
CAS No.:699020-93-4
- Rhapontisterone B
Catalog No.:BCN2664
CAS No.:698975-64-3
- (+)-Isoajmaline
Catalog No.:BCN3425
CAS No.:6989-79-3
- Evodine
Catalog No.:BCN2630
CAS No.:6989-38-4
- Bayogenin
Catalog No.:BCN2458
CAS No.:6989-24-8
- Atractylone
Catalog No.:BCN3048
CAS No.:6989-21-5
- Pseudoginsenoside F11
Catalog No.:BCN1062
CAS No.:69884-00-0
- Petunidin-3-O-glucoside chloride
Catalog No.:BCN3025
CAS No.:6988-81-4
- Boc-Glu(OtBu)-ONp
Catalog No.:BCC3393
CAS No.:69876-58-0
- Bourjotinolone A
Catalog No.:BCN4259
CAS No.:6985-35-9
- Neratinib (HKI-272)
Catalog No.:BCC3685
CAS No.:698387-09-6
- Swertisin
Catalog No.:BCN2762
CAS No.:6991-10-2
- 20(S),24(R)-Ocotillol
Catalog No.:BCN3891
CAS No.:69926-31-4
- Isocostic acid
Catalog No.:BCN4260
CAS No.:69978-82-1
- Triflurdine (Viroptic)
Catalog No.:BCC3873
CAS No.:70-00-8
- L-Glutathione Reduced
Catalog No.:BCC8030
CAS No.:70-18-8
- H-Asn-OH
Catalog No.:BCC2875
CAS No.:70-47-3
- 1-(4-Hydroxyphenyl)propan-1-one
Catalog No.:BCN4597
CAS No.:70-70-2
- H-Tyr(3-I)-OH
Catalog No.:BCC3265
CAS No.:70-78-0
- Indole-3-carbinol
Catalog No.:BCC5318
CAS No.:700-06-1
- 2-Adamantanol
Catalog No.:BCN8479
CAS No.:700-57-2
- 2-Adamantanone
Catalog No.:BCN8473
CAS No.:700-58-3
- Terazosin HCl
Catalog No.:BCC4354
CAS No.:70024-40-7
Characterization of a 5-HT1B receptor on CHO cells: functional responses in the absence of radioligand binding.[Pubmed:8882605]
Br J Pharmacol. 1996 Mar;117(6):1119-26.
1. Chinese hamster ovary (CHO) cells have been reported to be devoid of 5-HT receptors and have frequently been used as hosts for the expression of cloned 5-HT receptors. Unexpectedly, 5-HT was found to induce profound inhibition of forskolin-stimulated cyclic AMP production in these cells and the aim of this study was to classify the 5-HT receptor involved. 2. In CHO(dhfr-) cells 5-HT was a potent agonist and caused 80-100% inhibition of forskolin stimulated cyclic AMP production. A study using several 5-HT1 receptor agonists revealed the following potencies (p[A50]): RU24969 (9.09 +/- 0.17) > 5-carboxamidotryptamine (8.86 +/- 0.20) > 5-HT (8.07 +/- 0.05) > CP-93,129 (7.74 +/- 0.10) > sumatriptan (5.93 +/- 0.04). All five agonists achieved a similar maximum effect. Irreversible receptor alkylation studies yielded a pKA estimate of 7.04 +/- 0.34 for 5-HT. 3. The 5-HT1A/1B antagonist, (+/-)-cyanopindolol (4-100 nM), caused parallel rightward shifts of the 5-HT concentration-effect curve with no change in asymptote. Schild analysis yielded a pKB estimate of 8.69 +/- 0.09 (Schild slope 1.13 +/- 0.10). (+/-)-Cyanopindolol actually behaved as a partial agonist with an intrinsic activity of 0.2-0.5 and a p[A50] of 8.55. 4. 5-HT (0.01-10 microM) also elicited a concentration-dependent increase in intracellular [Ca2+] in CHO(dhfr-) cells thus demonstrating that dual coupling is not a phenomenon restricted to systems in which there is overexpression of transfected receptors. 5. This agonist and antagonist profile is consistent with the presence of a 5-HT1B receptor. 8-OH-DPAT (1 microM) and renzapride (3 microM) were without effect on forskolin-stimulated cyclic AMP production and ketanserin (0.3 microM) did not antagonize the inhibition produced by 5-HT, thus excluding the involvement of 5-HT1A, 5-HT4, and 5-HT2 receptors. 6. The possibility that expression of a 5-HT1B receptor was associated with the dhfr- mutation was excluded since RU24969, 5-HT and CP-93,129 were also potent agonists in unmutated, CHO-K1 cells: p[A50] 9.03 +/- 0.03, 8.34 +/- 0.05, 7.69 +/- 0.07 respectively, and (+/-)-cyanopindolol (0.1 microM) shifted the 5-HT curve to the right and yielded a pA2 estimate of 8.70 +/- 0.06. 7. Little or no specific binding of [3H]-5-HT (0.1-200 nM) or of the high affinity ligand [125I]-iodocyanopindolol (0.01-3 nM) to CHO(dhfr-) cell membranes could be detected. 5-HT also failed to elicit any increase in the binding of [35S]-GTP gamma S to CHO membranes. 8. In conclusion, cultured CHO cells express 5-HT1B receptors which are negatively coupled to adenylyl cyclase and positively coupled to increases in intracellular calcium. The absence of radioligand binding was unexpected in view of the high potency of 5-HT and the partial agonist activity of the normally 'silent' competitive antagonist, (+/-)-cyanopindolol. This implies very efficient receptor-effector coupling of a low density of 5-HT1B receptors. Clearly, the absence of detectable radioligand binding cannot be assumed to mean the absence of receptors capable of eliciting a significant functional response.
Atypical responses of rat ileum to pindolol, cyanopindolol and iodocyanopindolol.[Pubmed:8646418]
Br J Pharmacol. 1996 Feb;117(4):712-6.
1. Pindolol, cyanopindolol (CYP) and iodocyanopindolol (IodoCYP) have been reported to act either as antagonists, agonists or partial agonists at the beta 3-adrenoceptor in different preparations. A comprehensive investigation has not yet been described with these compounds tested in one tissue from one species. This study was conducted to delineate the pharmacological effects of pindolol, CYP and IodoCYP and to provide data on their affinities at the predominant beta-adrenoceptor in rat ileum. 2. The beta-adrenoceptors present in rat ileum were characterized in the presence of CGP 20712A and ICI 118 551, atropine and corticosterone, with (-)-isoprenaline used as an agonist. The role of the beta 1 and beta 2-adrenoceptors was determined by the omission of either CGP 20712A, ICI 118 551, or both, from the buffers. Conversely, the effectiveness of the beta 1- and beta 2-adrenoceptor blockade was examined by use of the beta 1-adrenoceptor-selective agonist, RO 363 and the beta 2-adrenoceptor-selective agonist, salbutamol. 3. There was no evidence for the presence of functional beta 1-adrenoceptors, and no strong evidence that beta 2-adrenoceptor stimulation contributed to the relaxant effects of (-)-isoprenaline. (-)-Phenylephrine did not produce relaxation of the tissue and 5-hydroxytryptamine produced contraction. 4. The beta 3-adrenoceptor-selective agonist, BRL 37344 and (-)-isoprenaline were potent full agonists (pD2 8.35 +/- 0.04 and 7.76 +/- 0.14 respectively), whereas ICI D7114 was less potent (pseudo pD2 6.92 +/- 0.15). These results indicate that the predominant functional beta-adrenoceptors in rat ileum are beta 3-adrenoceptors. 5. Partial agonist effects were produced by CYP (pD2 5.28 +/- 0.26) and IodoCYP (pD2 7.0 +/- 0.26), but not pindolol. All three compounds antagonized the effects of (-)-isoprenaline with pKb values of 6.68 +/- 0.10, 7.59 +/- 0.07 and 7.59 +/- 0.11 for pindolol, CYP and IodoCYP respectively. Likewise, CYP and IodoCYP antagonized the effects of BRL 37344 with pKb values of 7.20 +/- 0.22 and 7.21 +/- 0.14 respectively. This study provides the first functional data on the effects of IodoCYP, the ligand with the highest known affinity for the beta 3-adrenoceptor, at the characterized rat ileum beta 3-adrenoceptor. 6. In conclusion, whereas pKb values suggest that CYP and IodoCYP have a similar affinity for the beta 3-adrenoceptor in rat ileum, the higher potency of IodoCYP suggests that it promotes a greater coupling efficiency, or that its partial agonist effects are produced through a site other than the beta 3-adrenoceptor. The similar pKb values for CYP and IodoCYP at the beta 3-adrenoceptor contrast with their order of known affinities at the beta 1- and beta 2-adrenoceptors, where IodoCYP is far more potent than CYP. This provides evidence of further differences in the characteristics of the beta 3-adrenoceptors compared to the beta 1- and beta 2-adrenoceptors. Finally, the utility of IodoCYP as a beta 3-adrenoceptor antagonist would appear to be limited because of the greater magnitude of partial agonist effects that it produces.
Identity of inhibitory presynaptic 5-hydroxytryptamine (5-HT) autoreceptors in the rat brain cortex with 5-HT1B binding sites.[Pubmed:2936965]
Naunyn Schmiedebergs Arch Pharmacol. 1986 Jan;332(1):1-7.
In rat brain cortex slices preincubated with [3H]5-HT, the potencies of 17 5-HT receptor agonists to inhibit the electrically evoked 3H overflow and the affinities of 13 antagonists (including several beta-adrenoceptor blocking agents) to antagonize competitively the inhibitory effect of unlabelled 5-HT on evoked 3H overflow were determined. The affinities of the compounds for 5-HT1B and 5-HT2 binding sites in rat brain cortex membranes (labelled by [125I]cyanopindolol = [125I]-CYP in the presence of 30 mumol/l isoprenaline and [3H]ketanserin, respectively), for 5-HT1A binding sites in pig and rat brain cortex membranes (labelled by [3H]8-hydroxy-2-(di-n-propylamino)tetralin = [3H]8-OH-DPAT) and for 5-HT1C binding sites in pig choroid plexus membranes (labelled by [3H]mesulergine) were also determined. The affinities of the drugs for the various 5-HT recognition sites ranged over 4-5 log units (the functional experiments revealed the same range of differences between the drugs). There were no significant correlations between the affinities of the drugs at 5-HT1C and 5-HT2 binding sites and their potencies or affinities, determined for the 5-HT autoreceptors. In contrast, significant correlations were found between the potencies or affinities of the drugs for the autoreceptors and their affinities at 5-HT1A or 5-HT1B binding sites; the best correlations were obtained with the 5-HT1B binding site. Some of the drugs investigated were not included in the correlation since their agonistic or antagonistic effects on the autoreceptors were weak and pEC30 or apparent pA2 values could not be determined (less than 5.5).(ABSTRACT TRUNCATED AT 250 WORDS)


