L-AP6Selective agonist of the 'quis' effect CAS# 78944-89-5 |
- BMS-708163 (Avagacestat)
Catalog No.:BCC2104
CAS No.:1146699-66-2
- DAPT (GSI-IX)
Catalog No.:BCC3618
CAS No.:208255-80-5
- YO-01027 (Dibenzazepine, DBZ)
Catalog No.:BCC2100
CAS No.:209984-56-5
- Semagacestat (LY450139)
Catalog No.:BCC3610
CAS No.:425386-60-3
- Flurizan
Catalog No.:BCC2342
CAS No.:51543-40-9
- E 2012
Catalog No.:BCC1540
CAS No.:870843-42-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 78944-89-5 | SDF | Download SDF |
| PubChem ID | 2927 | Appearance | Powder |
| Formula | C6H14NO5P | M.Wt | 211.15 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in 1eq. NaOH | ||
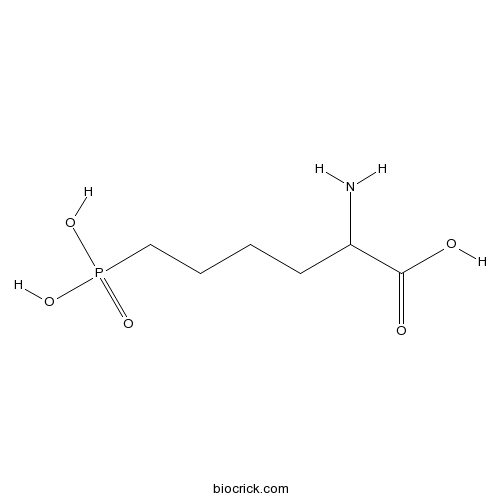
| Chemical Name | 2-amino-6-phosphonohexanoic acid | ||
| SMILES | C(CCP(=O)(O)O)CC(C(=O)O)N | ||
| Standard InChIKey | QIOXWRQXHFVNLV-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C6H14NO5P/c7-5(6(8)9)3-1-2-4-13(10,11)12/h5H,1-4,7H2,(H,8,9)(H2,10,11,12) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective agonist for 'quis'-sensitized site. |

L-AP6 Dilution Calculator

L-AP6 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.736 mL | 23.6798 mL | 47.3597 mL | 94.7194 mL | 118.3992 mL |
| 5 mM | 0.9472 mL | 4.736 mL | 9.4719 mL | 18.9439 mL | 23.6798 mL |
| 10 mM | 0.4736 mL | 2.368 mL | 4.736 mL | 9.4719 mL | 11.8399 mL |
| 50 mM | 0.0947 mL | 0.4736 mL | 0.9472 mL | 1.8944 mL | 2.368 mL |
| 100 mM | 0.0474 mL | 0.2368 mL | 0.4736 mL | 0.9472 mL | 1.184 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Chicanine
Catalog No.:BCN7818
CAS No.:78919-28-5
- Iloprost
Catalog No.:BCC7247
CAS No.:78919-13-8
- Deacetylnimbinene
Catalog No.:BCN4578
CAS No.:912545-53-0
- Orobanone
Catalog No.:BCN3562
CAS No.:78916-35-5
- 6-Thio-dG
Catalog No.:BCC6507
CAS No.:789-61-7
- 1-chloro-6-(5-ethynylthiophen-2-yl)hexa-3,5-diyn-2-ol
Catalog No.:BCN1351
CAS No.:78876-53-6
- 1-chloro-6-(5-(prop-1-ynyl)thiophen-2-yl)hexa-3,5-diyn-2-ol
Catalog No.:BCN1352
CAS No.:78876-52-5
- Demethylasterriquinone B1
Catalog No.:BCC7189
CAS No.:78860-34-1
- Garcinol
Catalog No.:BCC5623
CAS No.:78824-30-3
- Epibrassinolide
Catalog No.:BCC5479
CAS No.:78821-43-9
- 4-Benzyloxycarbonyl-2-piperazinone
Catalog No.:BCC8699
CAS No.:78818-15-2
- Zeylenol
Catalog No.:BCC8267
CAS No.:78804-17-8
- DL-AP7
Catalog No.:BCC6551
CAS No.:78966-69-5
- Guan-fu base G
Catalog No.:BCN8493
CAS No.:78969-72-9
- Methacrylamide
Catalog No.:BCN8157
CAS No.:79-39-0
- Oxytetracycline (Terramycin)
Catalog No.:BCC4819
CAS No.:79-57-2
- Lanosterol
Catalog No.:BCN3332
CAS No.:79-63-0
- Retinyl (Vitamin A) Palmitate
Catalog No.:BCC4749
CAS No.:79-81-2
- Camphene
Catalog No.:BCC9217
CAS No.:79-92-5
- 4'-O-Methylnyasol
Catalog No.:BCN7564
CAS No.:79004-25-4
- Eurycomalin A
Catalog No.:BCN3654
CAS No.:790234-20-7
- Masitinib (AB1010)
Catalog No.:BCC1260
CAS No.:790299-79-5
- SNAP
Catalog No.:BCC6712
CAS No.:79032-48-7
- β-Estradiol - d3
Catalog No.:BCC5365
CAS No.:79037-37-9
Utilization of the resolved L-isomer of 2-amino-6-phosphonohexanoic acid (L-AP6) as a selective agonist for a quisqualate-sensitized site in hippocampal CA1 pyramidal neurons.[Pubmed:7953634]
Brain Res. 1994 Jun 27;649(1-2):203-7.
Brief exposure of rat hippocampal slices to quisqualic acid (QUIS) sensitizes neurons to depolarization by the alpha-amino-omega-phosphonate excitatory amino acid (EAA) analogues AP4, AP5 and AP6. These phosphonates interact with a novel QUIS-sensitized site. Whereas L-AP4 and D-AP5 cross-react with other EAA receptors, DL-AP6 has been shown to be relatively selective for the QUIS-sensitized site. This specificity of DL-AP6, in conjunction with the apparent preference of this site for L-isomers, suggested that the hitherto unavailable L-isomer of AP6 would be a potent and specific agonist. We report the resolution of the D- and L-enantiomers of AP6 by fractional crystallization of the L-lysine salt of DL-AP6. We also report the pharmacological responses of kainate/AMPA, NMDA, lateral perforant path L-AP4 receptors and the CA1 QUIS-sensitized site to D- and L-AP6, and compare these responses to the D- and L-isomers of AP3, AP4, AP5 and AP7. The D-isomers of AP4, AP5 and AP6 were 5-, 3- and 14-fold less potent for the QUIS-sensitized site than their respective L-isomers. While L-AP4 and L-AP5 cross-reacted with NMDA and L-AP4 receptors, L-AP6 was found to be highly potent and specific for the QUIS-sensitized site (IC50 = 40 microM). Its IC50 values for kainate/AMPA, NMDA and L-AP4 receptors were > 10, 3 and 0.8 mM, respectively. As with AP4 and AP5, sensitization to L-AP6 was reversed by L-alpha-aminoadipate.
Type 4a metabotropic glutamate receptor: identification of new potent agonists and differentiation from the L-(+)-2-amino-4-phosphonobutanoic acid-sensitive receptor in the lateral perforant pathway in rats.[Pubmed:7623768]
Mol Pharmacol. 1995 Jul;48(1):140-9.
Before the discovery of the metabotropic glutamate receptors (mGluRs), the glutamate analogue L-2-amino-4-phosphonobutanoic acid (L-AP4) was identified as a potent presynaptic inhibitor of evoked synaptic transmission in the lateral perforant pathway in rats. The localization and L-AP4 sensitivity of the mGluR4a subtype of mGluRs were consistent with the hypothesis that this receptor mediates the synaptic depressant effects of L-AP4 in the lateral perforant pathway. In the present study, the pharmacology of mGluR4a expressed in baby hamster kidney 570 cells was characterized and compared with that previously reported for the lateral perforant pathway responses. The endogenous excitatory amino acid L-aspartate was inactive at mGluR4a, whereas L-homocysteic acid was only 5-fold less potent than L-glutamate. These data suggest that L-homocysteic acid may be an endogenous agonist at mGluR4a. Of the 30 L-AP4 analogues examined, several compounds were identified as agonists at mGluR4a. The cyclopropyl-AP4 analogue (Z)-(+/-)-2-amino-2,3-methano-4-phosphonobutanoic acid inhibited forskolin-stimulated cAMP production with an EC50 of 0.58 microM, which is comparable to that of L-AP4 (EC50 = 0.43 microM). Two other cyclic analogues of L-AP4 were approximately 10-fold less potent as agonists at mGluR4a, i.e., (+/-)-1-amino-3-(phosphonomethylene)cyclobutanecarboxylic acid (EC50 = 4.4 microM) and (E)-(+/-)-2-amino-2,3-methano-4-phosphonobutanoic acid (EC50 = 7.9 microM). Comparison of the potencies of the compounds for activation of mGluR4a with their potencies for inhibition of lateral perforant pathway responses demonstrates that some compounds have comparable activities in the two systems, whereas several compounds are at least 10-fold more potent in one of the systems. In addition, although the mGluR antagonist (+)-alpha-methyl-4-carboxyphenylglycine blocked the effects of L-AP4 in the lateral perforant pathway, it did not block the effects of L-AP4 at the cloned receptor. These data provide evidence that mGluR4a does not mediate the effects of L-AP4 in the lateral perforant pathway, they provide new tools to identify the function of these receptors in the mammalian central nervous system, and they indicate that the effects of L-AP4 in the lateral perforant pathway are mediated by a (+)-alpha-methyl-4-carboxyphenylglycine-sensitive receptor.
Synthesis of oxadiazolidinedione derivatives as quisqualic acid analogues and their evaluation at a quisqualate-sensitized site in the rat hippocampus.[Pubmed:7966155]
J Med Chem. 1994 Nov 11;37(23):3939-46.
The ability of quisqualic acid (1) to sensitize neurons to depolarization by omega-phosphono alpha-amino acid analogues of excitatory amino acids is a highly specific phenomenon and is termed the QUIS effect. In an attempt to elucidate the structure-activity relationships for this sensitization, analogues 2-6 of quisqualic acid have been synthesized. Compounds 4, 5, and 6 showed no quisqualate sensitization with respect to L-2-amino-6-phosphonohexanoic acid (L-AP6), while compounds 2 and 3 were 1/10 and 1/1000, respectively, as active as quisqualic acid in sensitizing neurons toward L-AP6.


