Lauric acidCAS# 143-07-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 143-07-7 | SDF | Download SDF |
| PubChem ID | 3893 | Appearance | Powder |
| Formula | C12H24O2 | M.Wt | 200.31 |
| Type of Compound | Miscellaneous | Storage | Desiccate at -20°C |
| Solubility | DMSO : ≥ 250 mg/mL (1248.00 mM) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | dodecanoic acid | ||
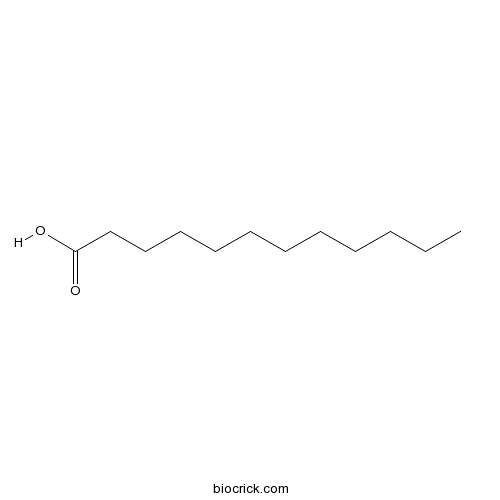
| SMILES | CCCCCCCCCCCC(=O)O | ||
| Standard InChIKey | POULHZVOKOAJMA-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C12H24O2/c1-2-3-4-5-6-7-8-9-10-11-12(13)14/h2-11H2,1H3,(H,13,14) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Lauric acid is a middle chain-free fatty acid with strong bactericidal properties. The EC50s for P. acnes, S.aureus, S. epidermidis, are 2, 6, 4 μg/mL, respectively. Lauric acid displays preferential antineoplastic property, including induction of apoptosis in a CRC cell line. |
| Targets | IL Receptor | TNF-α | NF-kB | ROS | MAPK |
| In vitro | Lauric acid in crown daisy root exudate potently regulates root-knot nematode chemotaxis and disrupts Mi-flp-18 expression to block infection.[Pubmed: 24170741]J Exp Bot. 2014 Jan;65(1):131-41.Tomato (Solanum lycopersicum) crops can be severely damaged due to parasitism by the root-knot nematode (RKN) Meloidogyne incognita, but are protected when intercropped with crown daisy (Chrysanthemum coronarium L.). Root exudate may be the determining factor for this protection. |
| In vivo | Anti-bacterial and anti-inflammatory properties of capric acid against Propionibacterium acnes: a comparative study with lauric acid.[Pubmed: 24284257]J Dermatol Sci. 2014 Mar;73(3):232-40.Propionibacterium acnes (P. acnes) is a commensal bacterium which is possibly involved in acne inflammation. The saturated fatty acid, Lauric acid (C12:0) has been shown to possess antibacterial and anti-inflammatory properties against P. acnes. Little is known concerning the potential effects of its decanoic counterpart, capric acid (C10:0).
To examine the antibacterial and anti-inflammatory activities of capric acid against P. acnes and to investigate the mechanism of the anti-inflammatory action.
|
| Cell Research | Induction of apoptosis by the medium-chain length fatty acid lauric acid in colon cancer cells due to induction of oxidative stress.[Pubmed: 24356281]Chemotherapy. 2013;59(3):214-24.Fatty acids are classified as short-chain (SCFA), medium-chain (MCFA) or long-chain and hold promise as adjunctive chemotherapeutic agents for the treatment of colorectal cancer. The antineoplastic potential of MCFA remains underexplored; accordingly, we compared the MCFA Lauric acid (C12:0) to the SCFA butyrate (C4:0) in terms of their capacity to induce apoptosis, modify glutathione (GSH) levels, generate reactive oxygen species (ROS), and modify phases of the cell cycle in Caco-2 and IEC-6 intestinal cell lines.
|

Lauric acid Dilution Calculator

Lauric acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.9923 mL | 24.9613 mL | 49.9226 mL | 99.8452 mL | 124.8065 mL |
| 5 mM | 0.9985 mL | 4.9923 mL | 9.9845 mL | 19.969 mL | 24.9613 mL |
| 10 mM | 0.4992 mL | 2.4961 mL | 4.9923 mL | 9.9845 mL | 12.4807 mL |
| 50 mM | 0.0998 mL | 0.4992 mL | 0.9985 mL | 1.9969 mL | 2.4961 mL |
| 100 mM | 0.0499 mL | 0.2496 mL | 0.4992 mL | 0.9985 mL | 1.2481 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Salvianolic acid D
Catalog No.:BCN2369
CAS No.:142998-47-8
- Salvianolic acid E
Catalog No.:BCN8194
CAS No.:142998-46-7
- Fmoc-D-Ala(3-pyridyl)-OH.HCl
Catalog No.:BCC3324
CAS No.:142994-45-4
- Fmoc-D-Phe(4-Cl)-OH
Catalog No.:BCC3177
CAS No.:142994-19-2
- UNC2025
Catalog No.:BCC8062
CAS No.:1429881-91-3
- HPOB
Catalog No.:BCC5574
CAS No.:1429651-50-2
- Triptoquinone A
Catalog No.:BCN6781
CAS No.:142950-86-5
- Triptoquinone B
Catalog No.:BCN6238
CAS No.:142937-50-6
- Mutant IDH1 inhibitor
Catalog No.:BCC4144
CAS No.:1429180-08-4
- OXF BD 02
Catalog No.:BCC5598
CAS No.:1429129-68-9
- Petunidin chloride
Catalog No.:BCN3018
CAS No.:1429-30-7
- 8alpha-Methacryloyloxy-13-ethoxyvernojalcanolide
Catalog No.:BCN7445
CAS No.:142891-14-3
- Protoveratrine A
Catalog No.:BCN5346
CAS No.:143-57-7
- Vinblastine Sulfate
Catalog No.:BCN2292
CAS No.:143-67-9
- ML216
Catalog No.:BCC8061
CAS No.:1430213-30-1
- 1alpha-Hydroxy VD4
Catalog No.:BCC1300
CAS No.:143032-85-3
- Ethyl 3,4-dicaffeoylquinate
Catalog No.:BCN8004
CAS No.:143051-73-4
- Phytic acid sodium salt hydrate
Catalog No.:BCN1283
CAS No.:14306-25-3
- BQ-3020
Catalog No.:BCC5728
CAS No.:143113-45-5
- Neotuberostemonine
Catalog No.:BCN6239
CAS No.:143120-46-1
- GSK-LSD1 2HCl
Catalog No.:BCC5647
CAS No.:1431368-48-7
- BMX-IN-1
Catalog No.:BCC1434
CAS No.:1431525-23-3
- H-D-Ala-OMe.HCl
Catalog No.:BCC3199
CAS No.:14316-06-4
- UNC1999
Catalog No.:BCC4552
CAS No.:1431612-23-5
Induction of apoptosis by the medium-chain length fatty acid lauric acid in colon cancer cells due to induction of oxidative stress.[Pubmed:24356281]
Chemotherapy. 2013;59(3):214-24.
BACKGROUND: Fatty acids are classified as short-chain (SCFA), medium-chain (MCFA) or long-chain and hold promise as adjunctive chemotherapeutic agents for the treatment of colorectal cancer. The antineoplastic potential of MCFA remains underexplored; accordingly, we compared the MCFA Lauric acid (C12:0) to the SCFA butyrate (C4:0) in terms of their capacity to induce apoptosis, modify glutathione (GSH) levels, generate reactive oxygen species (ROS), and modify phases of the cell cycle in Caco-2 and IEC-6 intestinal cell lines. METHODS: Caco-2 and IEC-6 cells were treated with Lauric acid, butyrate, or vehicle controls. Apoptosis, ROS, and cell cycle analysis were determined by flow cytometry. GSH availability was assessed by enzymology. RESULTS: Lauric acid induced apoptosis in Caco-2 (p < 0.05) and IEC-6 cells (p < 0.05) compared to butyrate. In Caco-2 cells, Lauric acid reduced GSH availability and generated ROS compared to butyrate (p < 0.05). Lauric acid reduced Caco-2 and IEC-6 cells in G0/G1and arrested cells in the S and G2/M phases. Lauric acid induced apoptosis in IEC-6 cells compared to butyrate (p < 0.05). Butyrate protected IEC-6 cells from ROS-induced damage, whereas Lauric acid induced high levels of ROS compared to butyrate. CONCLUSION: Compared to butyrate, Lauric acid displayed preferential antineoplastic properties, including induction of apoptosis in a CRC cell line.
Lauric acid in crown daisy root exudate potently regulates root-knot nematode chemotaxis and disrupts Mi-flp-18 expression to block infection.[Pubmed:24170741]
J Exp Bot. 2014 Jan;65(1):131-41.
Tomato (Solanum lycopersicum) crops can be severely damaged due to parasitism by the root-knot nematode (RKN) Meloidogyne incognita, but are protected when intercropped with crown daisy (Chrysanthemum coronarium L.). Root exudate may be the determining factor for this protection. An experiment using pots linked by a tube and Petri dish experiments were undertaken to confirm that tomato-crown daisy intercropping root exudate decreased the number of nematodes and alleviated nematode damage, and to determine crown daisy root exudate-regulated nematode chemotaxis. Following a gas chromatography-mass spectrometry assay, it was found that the intercropping protection was derived from the potent bioactivity of a specific root exudate component of crown daisy, namely Lauric acid. The Mi-flp-18 gene, encoding an FMRFamide-like peptide neuromodulator, regulated nematode chemotaxis and infection by RNA interference. Moreover, it was shown that Lauric acid acts as both a lethal trap and a repellent for M. incognita by specifically regulating Mi-flp-18 expression in a concentration-dependent manner. Low concentrations of Lauric acid (0.5-2.0mM) attract M. incognita and consequently cause death, while high concentrations (4.0mM) repel M. incognita. This study elucidates how Lauric acid in crown daisy root exudate regulates nematode chemotaxis and disrupts Mi-flp-18 expression to alleviate nematode damage, and presents a general methodology for studying signalling systems affected by plant root exudates in the rhizosphere. This could lead to the development of economical and feasible strategies for controlling plant-parasitic nematodes, and provide an alternative to the use of pesticides in farming systems.
Anti-bacterial and anti-inflammatory properties of capric acid against Propionibacterium acnes: a comparative study with lauric acid.[Pubmed:24284257]
J Dermatol Sci. 2014 Mar;73(3):232-40.
BACKGROUND: Propionibacterium acnes (P. acnes) is a commensal bacterium which is possibly involved in acne inflammation. The saturated fatty acid, Lauric acid (C12:0) has been shown to possess antibacterial and anti-inflammatory properties against P. acnes. Little is known concerning the potential effects of its decanoic counterpart, capric acid (C10:0). OBJECTIVE: To examine the antibacterial and anti-inflammatory activities of capric acid against P. acnes and to investigate the mechanism of the anti-inflammatory action. METHODS: The antimicrobial activity of fatty acids was detected using the broth dilution method. An evaluation of P. acnes-induced ear edema in mice was conducted to evaluate the in vivo anti-inflammatory effect. To elucidate the in vitro anti-inflammatory effect, human SZ95 sebocytes and monocytic THP-1 cells were treated with P. acnes alone or in the presence of a fatty acid. The mRNA levels and secretion of pro-inflammatory cytokines were measured by qRT-PCR and enzyme immunoassay, respectively. NF-kappaB activation and MAPK expression were analyzed by ELISA and Western blot, respectively. RESULTS: Lauric acid had stronger antimicrobial activity against P. acnes than capric acid in vitro and in vivo. However, both fatty acids attenuated P. acnes-induced ear swelling in mice along with microabscess and significantly reduced interleukin (IL)-6 and CXCL8 (also known as IL-8) production in P. acnes-stimulated SZ95 sebocytes. P. acnes-induced mRNA levels and secretion of IL-8 and TNF-alpha in THP-1 cells were suppressed by both fatty acids, which inhibited NF-kappaB activation and the phosphorylation of MAP kinases. CONCLUSION: Our data demonstrate that both capric acid and Lauric acid exert bactericidal and anti-inflammatory activities against P. acnes. The anti-inflammatory effect may partially occur through the inhibition of NF-kappaB activation and the phosphorylation of MAP kinases.
Controlling methanogenesis and improving power production of microbial fuel cell by lauric acid dosing.[Pubmed:25353941]
Water Sci Technol. 2014;70(8):1363-9.
Methanogens compete with anodophiles for substrate and thus reduce the power generation and coulombic efficiency (CE) of the microbial fuel cell (MFC). Performance of a baked clayware membrane MFC inoculated with mixed anaerobic sludge pretreated with Lauric acid was investigated in order to enhance power recovery by controlling methanogenesis. In the presence of Lauric acid pretreated inoculum, MFC produced maximum volumetric power density of 4.8 W/m(3) and the CE increased from 3.6% (for untreated inoculum) to 11.6%. Cyclic voltammetry (CV) and electro-kinetic evaluation indicated a higher bio-catalytic activity at the anode of the MFC inoculated with Lauric acid pretreated sludge. With the Lauric acid pretreated inoculum a higher catalytic current of 114 mA, exchange current density of 40.78 mA/m(2) and lower charge transfer resistance of 0.00016 Omegam(2) were observed during oxidation at the anode. Addition of Lauric acid significantly achieved suppression of methanogenesis and enhanced the sustainable power generation of MFC by 3.9 times as compared with control MFC inoculated with sludge without any pretreatment.


