PhyltetralinCAS# 123048-17-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 123048-17-9 | SDF | Download SDF |
| PubChem ID | 11223782 | Appearance | Powder |
| Formula | C24H32O6 | M.Wt | 416.5 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
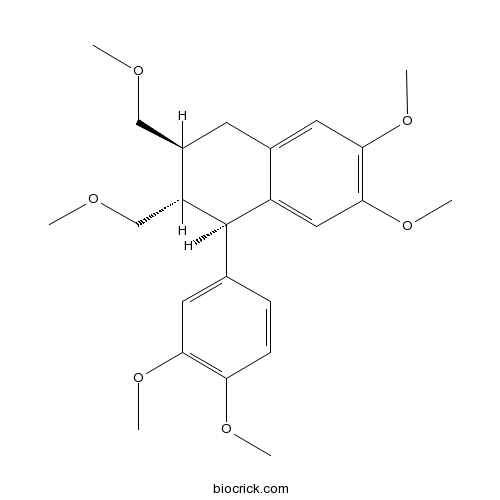
| Chemical Name | (1R,2S,3S)-1-(3,4-dimethoxyphenyl)-6,7-dimethoxy-2,3-bis(methoxymethyl)-1,2,3,4-tetrahydronaphthalene | ||
| SMILES | COCC1CC2=CC(=C(C=C2C(C1COC)C3=CC(=C(C=C3)OC)OC)OC)OC | ||
| Standard InChIKey | CZZKSEXMNQGXJU-ROMRWMGNSA-N | ||
| Standard InChI | InChI=1S/C24H32O6/c1-25-13-17-9-16-11-22(29-5)23(30-6)12-18(16)24(19(17)14-26-2)15-7-8-20(27-3)21(10-15)28-4/h7-8,10-12,17,19,24H,9,13-14H2,1-6H3/t17-,19-,24-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Phyltetralin has antihyperuricemic activity. |
| Targets | Immunology & Inflammation related |

Phyltetralin Dilution Calculator

Phyltetralin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.401 mL | 12.0048 mL | 24.0096 mL | 48.0192 mL | 60.024 mL |
| 5 mM | 0.4802 mL | 2.401 mL | 4.8019 mL | 9.6038 mL | 12.0048 mL |
| 10 mM | 0.2401 mL | 1.2005 mL | 2.401 mL | 4.8019 mL | 6.0024 mL |
| 50 mM | 0.048 mL | 0.2401 mL | 0.4802 mL | 0.9604 mL | 1.2005 mL |
| 100 mM | 0.024 mL | 0.12 mL | 0.2401 mL | 0.4802 mL | 0.6002 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Bulleyanin
Catalog No.:BCN6120
CAS No.:123043-54-9
- Azasetron HCl
Catalog No.:BCC5035
CAS No.:123040-16-4
- Azelaic Acid
Catalog No.:BCC8300
CAS No.:123-99-9
- D-erythro-Sphingosine (synthetic)
Catalog No.:BCC6729
CAS No.:123-78-4
- Anisic aldehyde
Catalog No.:BCN2618
CAS No.:123-11-5
- 4-Hydroxybenzaldehyde
Catalog No.:BCN5816
CAS No.:123-08-0
- Fmoc-Hyp(tBu)-OH
Catalog No.:BCC3256
CAS No.:122996-47-8
- RG7388
Catalog No.:BCC1895
CAS No.:1229705-06-9
- HA 130
Catalog No.:BCC7884
CAS No.:1229652-21-4
- 6'-O-Galloyl paeoniflorin
Catalog No.:BCN2941
CAS No.:122965-41-7
- URMC-099
Catalog No.:BCC5563
CAS No.:1229582-33-5
- XE 991 dihydrochloride
Catalog No.:BCC7232
CAS No.:122955-13-9
- BAF312 (Siponimod)
Catalog No.:BCC5114
CAS No.:1230487-00-9
- 4,8-Dihydroxyeudesm-7(11)-en-12,8-olide
Catalog No.:BCN1600
CAS No.:1231208-53-9
- Uncaric acid
Catalog No.:BCN6121
CAS No.:123135-05-7
- Cefepime Dihydrochloride Monohydrate
Catalog No.:BCC5261
CAS No.:123171-59-5
- LY2835219 free base
Catalog No.:BCC1722
CAS No.:1231929-97-7
- LY2835219
Catalog No.:BCC1113
CAS No.:1231930-82-7
- VE-821
Catalog No.:BCC1207
CAS No.:1232410-49-9
- 6alpha-Hydroxytomentosin
Catalog No.:BCN7303
CAS No.:1232676-22-0
- 2'-O-Methylhelichrysetin
Catalog No.:BCN4792
CAS No.:123316-64-3
- Clofarabine
Catalog No.:BCC1078
CAS No.:123318-82-1
- AZ20
Catalog No.:BCC1389
CAS No.:1233339-22-4
- Dipsacobioside
Catalog No.:BCN6552
CAS No.:123350-57-2
Anti-oxidant and inflammatory mediator's growth inhibitory effects of compounds isolated from Phyllanthus urinaria.[Pubmed:18187278]
J Ethnopharmacol. 2008 Mar 5;116(2):333-40.
Phyllanthus urinaria Linnea (Euphorbiaceae), is a traditional anti-hepatitis herb used in Taiwan. In continuation of our search for potent natural anti-inflammatory agents, from the ethanolic extract of this plant, nine compounds including phyllanthin (1), Phyltetralin (2), trimethyl-3,4-dehydrochebulate (3), methylgallate (4), and rhamnocitrin (5), methyl brevifolincarboxylate (6), beta-sitosterol-3-O-beta-d-glucopyranoside (7), quercitrin (8), and rutin (9) were isolated. The structures of compounds 3 and 6 were established based on NMR and mass spectral studies. The isolates 1-9 were investigated for their antioxidant, and anti-inflammatory activities in vitro. In the antioxidant assay, the isolates 3, 4 and 6 exhibited significant DPPH radical scavenging activity with an IC(50) value of 9.4, 9.8 and 8.9 microM, respectively. On the other hand, in the inflammatory mediators growth inhibitory assay from LPS/interferon (IFN)-gamma-activated peritoneal macrophages, all the isolates except 7, significantly and dose-dependently inhibited the enhanced production of NO radicals, and such modulation was closely associated with the inhibition of tumor necrosis factor (TNF)-alpha and interleukin (IL)-6. In addition, 30 microM of isolates 3 and 6, and 50 microM of 4, significantly arrest the mitogen-stimulated spleen cells in G0/G1 stage. This is the first report on Phyllanthus urinaria isolates for their growth inhibitory activities against inflammatory mediators, in addition to spleen cell cycle arrest in G0/G1 stage. Therefore, these isolates from Phyllanthus urinaria may be useful for the treatment of cell-mediated immune diseases.
The cytotoxic effect and the multidrug resistance reversing action of lignans from Phyllanthus amarus.[Pubmed:17054045]
Planta Med. 2006 Dec;72(15):1353-8.
Multidrug resistance (MDR) constitutes the major obstacle to the successful treatment of cancer. In several cancer cells, MDR is thought to be mediated by the super-expression of P-glycoprotein (Pgp). Pgp extrudes drugs from the cells, therefore reducing their cytotoxicity, and its activity inhibition may reverse the MDR phenotype. The present study evaluated the possible cytotoxic effect and MDR reversing properties of the extract and compounds isolated from Phyllanthus amarus. To this purpose, two human leukaemia cell lines were employed: K-562 and its vincristine-resistant counterpart Lucena-1, a Pgp-overexpressing subline. We report here that Lucena-1 was significantly more resistant to the cytotoxicity of P. amarus derivatives: the hexane extract (HE, 100 microg/mL), the lignans-rich fraction (LRF, 100 microg/mL) and the lignans nirtetralin (NIRT, 43.2 microg/mL), niranthin (NIRA, 43 microg/mL) or phyllanthin (PHYLLA, 43 microg/mL) exerted cytotoxic effects on K-562 cells with 40.3, 66.0, 62.0, 61.0 or 24.1% of cell death, respectively. The cellular toxicity observed on Lucena-1 was 16.3, 40.4, 29.4, 30.2, or 24.8%, respectively. However, cell treatment with the lignan Phyltetralin (PHYLT) up to 41.6 microg/mL had no cytotoxic action on either of the cell lines. P. amarus derivatives were also found to be effective in inhibiting Pgp activity as assessed by rhodamine accumulation in Lucena-1 cells, as were the classical Pgp inhibitors, cyclosporine A (160 nM), PSC-833 (2 microM) and verapamil (5 microM). The lignan NIRT produced the most potent inhibition (EC (50) = 29.4 microg/mL) followed by NIRA (44.3 microg/mL), LRF (49.1 microg/mL), PHYLT (99.4 microg/mL), PHYLLA and HE (> 100 microg/mL). Lucena-1 cells were more resistant to daunorubicin-induced cell death (LC (50) = 50 microM) than K562 cells (LC (50) = 4.95 microM). Of note, the P. amarus derivatives significantly potentiated 5 microM daunorubicin-induced cell death in Lucena-1 cells (P < 0.01) but not in K562 cells. After treatment only with P. amarus derivatives (100 microg/mL HE, 30 microg/mL LRF, 12.9 microg/mL NIRA, 43.2 microg/mL NIRT, 43 microg/mL PHYLLA or 41.6 microg/mL PHYLT), the Lucena-1 cellular viability was 83.7, 85.3, 101, 69.7, 75.6 or 88.7%, respectively, whereas the in the presence of daunorubincin, which was not cytotoxic PER SE, the cell viability decreased to 42.9, 42.2, 64.2, 35.4, 30.4 or 52.6%, respectively. Together, these results suggest a potential action of P. amarus derivatives as MDR reversing agents, mainly due to their ability to synergize with the action of conventional chemotherapeutics.
Antihyperuricemic lignans from the leaves of Phyllanthus niruri.[Pubmed:16953466]
Planta Med. 2006 Nov;72(14):1262-7.
The methanol extract from the leaves of Phyllanthus niruri L. showed oral antihyperuricemic activity in potassium oxonate- and uric acid-induced hyperuricemic rats. Fractionation of the extract by resin chromatography led to the isolation of a less polar fraction which exhibited the highest reduction of plasma uric acid. Further antihyperuricemic-guided purification of the fraction afforded three lignans, phyllanthin (1), hypophyllanthin (2) and Phyltetralin (3), of which 1 significantly reversed the plasma uric acid level of hyperuricemic animals to its normal level in a dose-dependent manner, comparable to that of allopurinol, benzbromarone and probenecid which are used clinically for the treatment of hyperuricemia and gout. Thus, the lignans of P. niruri are potential antihyperuricemic agents worthy of further investigation.


