Pre-schisanartanin BCAS# 1033288-92-4 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1033288-92-4 | SDF | Download SDF |
| PubChem ID | 124865186 | Appearance | Cryst. |
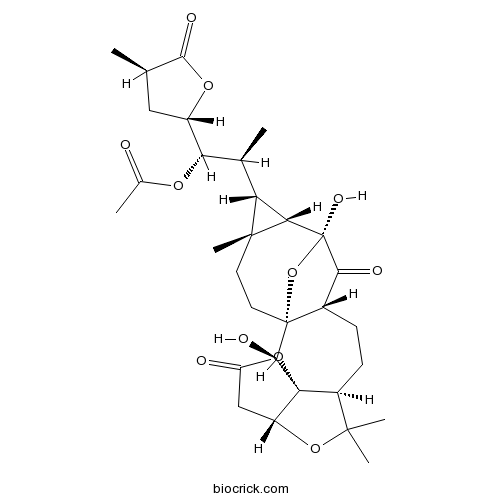
| Formula | C31H42O11 | M.Wt | 590.7 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | [(1S,2S)-2-[(1R,2S,3S,7R,10S,13R,15S,16S,17R,18R)-2,15-dihydroxy-9,9,18-trimethyl-5,14-dioxo-4,8,21-trioxahexacyclo[13.5.1.01,13.03,7.03,10.016,18]henicosan-17-yl]-1-[(2S,4R)-4-methyl-5-oxooxolan-2-yl]propyl] acetate | ||
| SMILES | CC1CC(OC1=O)C(C(C)C2C3C2(CCC45C(CCC6C(OC7C6(C4O)OC(=O)C7)(C)C)C(=O)C3(O5)O)C)OC(=O)C | ||
| Standard InChIKey | XVOAOTAZULSEBL-UAALQDDJSA-N | ||
| Standard InChI | InChI=1S/C31H42O11/c1-13-11-17(39-25(13)35)22(38-15(3)32)14(2)21-23-28(21,6)9-10-29-16(24(34)31(23,37)42-29)7-8-18-27(4,5)40-19-12-20(33)41-30(18,19)26(29)36/h13-14,16-19,21-23,26,36-37H,7-12H2,1-6H3/t13-,14+,16+,17+,18+,19-,21-,22+,23+,26+,28-,29-,30-,31+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Pre-schisanartanin B has anti-HIV-1 activity. 2. Pre-schisanartanin B has cytotoxicity. |
| Targets | HIV |

Pre-schisanartanin B Dilution Calculator

Pre-schisanartanin B Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.6929 mL | 8.4645 mL | 16.9291 mL | 33.8581 mL | 42.3227 mL |
| 5 mM | 0.3386 mL | 1.6929 mL | 3.3858 mL | 6.7716 mL | 8.4645 mL |
| 10 mM | 0.1693 mL | 0.8465 mL | 1.6929 mL | 3.3858 mL | 4.2323 mL |
| 50 mM | 0.0339 mL | 0.1693 mL | 0.3386 mL | 0.6772 mL | 0.8465 mL |
| 100 mM | 0.0169 mL | 0.0846 mL | 0.1693 mL | 0.3386 mL | 0.4232 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Taltirelin
Catalog No.:BCC5271
CAS No.:103300-74-9
- LDK378
Catalog No.:BCC3691
CAS No.:1032900-25-6
- GSK1292263
Catalog No.:BCC3786
CAS No.:1032823-75-8
- PTIQ
Catalog No.:BCC7953
CAS No.:1032822-42-6
- GDC-0980 (RG7422)
Catalog No.:BCC4992
CAS No.:1032754-93-0
- GNE-477
Catalog No.:BCC8049
CAS No.:1032754-81-6
- BAY 80-6946 (Copanlisib)
Catalog No.:BCC4986
CAS No.:1032568-63-0
- L-655,240
Catalog No.:BCC7156
CAS No.:103253-15-2
- MK-2206 dihydrochloride
Catalog No.:BCC1274
CAS No.:1032350-13-2
- D-Arabinose
Catalog No.:BCN3791
CAS No.:10323-20-3
- A939572
Catalog No.:BCC5305
CAS No.:1032229-33-6
- Fmoc-Cys(Trt)-OH
Catalog No.:BCC3479
CAS No.:103213-32-7
- 3-Oxo-4-aza-5-alpha-androstane-17β-carboxylic acid
Catalog No.:BCC8641
CAS No.:103335-55-3
- L-364,373
Catalog No.:BCC7445
CAS No.:103342-82-1
- 1-Methyl-L-4,5-dihydroorotic acid
Catalog No.:BCC8472
CAS No.:103365-69-1
- GNE-493
Catalog No.:BCC8048
CAS No.:1033735-94-2
- Itol A
Catalog No.:BCN5847
CAS No.:1033747-78-2
- GNF-5837
Catalog No.:BCC3668
CAS No.:1033769-28-6
- Salidroside
Catalog No.:BCN5966
CAS No.:10338-51-9
- Telotristat
Catalog No.:BCC5128
CAS No.:1033805-28-5
- HPGDS inhibitor 1
Catalog No.:BCC4065
CAS No.:1033836-12-2
- 1-O-Methylnataloe-emodin
Catalog No.:BCN7036
CAS No.:103392-51-4
- Octyl gallate
Catalog No.:BCN8432
CAS No.:1034-01-1
- Disodium (R)-2-Hydroxyglutarate
Catalog No.:BCC6515
CAS No.:103404-90-6
Isolation and characterization of miscellaneous terpenoids of Schisandra chinensis
Tetrahedron Volume 64, Issue 19, 5 May 2008, Pages 4260–4267
Two new bisnortriterpenoids with 18-norschiartane skeleton, wuweizidilactones G (1) and H (2), four new highly oxygenated nortriterpenoids based on a schisanartane skeleton, schindilactones D–G (3–6), a pre-schisanartane skeleton, Pre-schisanartanin B (7), and a novel 3,4-seco-21,26-olide-artane triterpenoid wuweizilactone acid (8), along with 24 known terpenoids with different carbon frameworks, have been isolated from the acetone extract of the stems and leaves of Schisandra chinensis. The terpenoids produced by this plant have chemical diversity. The structures of new compounds 1–8 have been characterized by spectroscopic data interpretation. The cytotoxicity and anti-HIV-1 activity of all the Schisandra nortriterpenoids were evaluated.


