XanthotoxinCAS# 298-81-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 298-81-7 | SDF | Download SDF |
| PubChem ID | 4114 | Appearance | White/cream powder |
| Formula | C12H8O4 | M.Wt | 216.2 |
| Type of Compound | Coumarins | Storage | Desiccate at -20°C |
| Synonyms | 8-Methoxypsoralen | ||
| Solubility | DMSO : 50 mg/mL (231.28 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
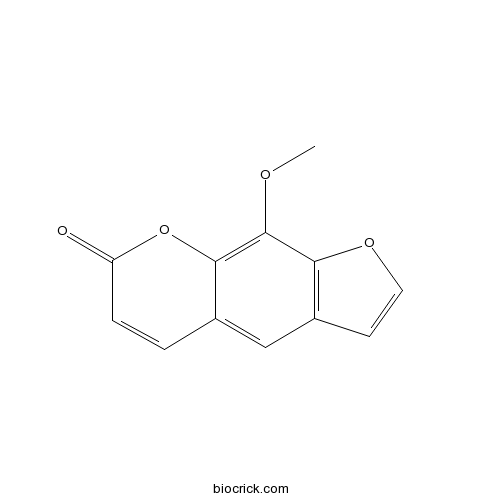
| Chemical Name | 9-methoxyfuro[3,2-g]chromen-7-one | ||
| SMILES | COC1=C2C(=CC3=C1OC=C3)C=CC(=O)O2 | ||
| Standard InChIKey | QXKHYNVANLEOEG-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C12H8O4/c1-14-12-10-8(4-5-15-10)6-7-2-3-9(13)16-11(7)12/h2-6H,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Xanthotoxin (Methoxsalen ) is a potent tricyclic furocoumarin suicide inhibitor of CYP (cytochrome P-450), is an agent used to treat psoriasis, eczema, vitiligo and some cutaneous Lymphomas in conjunction with exposing the skin to sunlight. Xanthotoxin has anticonvulsant activities, it can protect the animals against maximal electroshock-induced seizures; Xanthotoxin prevents bone loss in ovariectomized mice through the inhibition of RANKL-induced osteoclastogenesis, it may be considered to be a new therapeutic candidate for treating osteoporosis. |
| Targets | FAK | ROS | Calcium Channel | P450 (e.g. CYP17) | NFATc1 | c-FOS |
| In vitro | Xanthotoxin prevents bone loss in ovariectomized mice through the inhibition of RANKL-induced osteoclastogenesis.[Pubmed: 26809192]Osteoporos Int. 2016 Jul;27(7):2335-2344.Xanthotoxin (XAT) is extracted from the seeds of Ammi majus. Here, we reported that XAT has an inhibitory effect on osteoclastogenesis in vitro through the suppression of both receptor activator of nuclear factor-κB ligand (RANKL)-induced ROS generation and Ca(2+) oscillations. In vivo studies showed that XAT treatment decreases the osteoclast number, prevents bone loss, and restores bone strength in ovariectomized mice.
Excessive osteoclast formation and the resultant increase in bone resorption activity are key pathogenic factors of osteoporosis. In the present study, we have investigated the effects of XAT, a natural furanocoumarin, on the RANKL-mediated osteoclastogenesis in vitro and on ovariectomy-mediated bone loss in vivo.
|
| In vivo | Effects of methoxsalen, a CYP2A5/6 inhibitor, on nicotine dependence behaviors in mice.[Pubmed: 24859605]Neuropharmacology. 2014 Oct;85:67-72.Metabolism of nicotine to inactive cotinine by hepatic enzyme CYP2A6 is the principal pathway by which active nicotine is removed from circulation.
Anticonvulsant effects of four linear furanocoumarins, bergapten, imperatorin, oxypeucedanin, and xanthotoxin, in the mouse maximal electroshock-induced seizure model: a comparative study.[Pubmed: 21273683]Pharmacol Rep. 2010 Nov-Dec;62(6):1231-6.The aim of this study was to determine and compare the anticonvulsant activities of four natural furanocoumarins [bergapten (5-methoxypsoralen), imperatorin (8-isopentenyloxypsoralen), oxypeucedanin (5-epoxy-isopentenyloxypsoralen) and Xanthotoxin (8-methoxypsoralen)] in the maximal electroshock-induced seizure test in mice.
|
| Kinase Assay | Effect of coumarin and xanthotoxin on mitochondrial structure, oxygen uptake, and succinate dehydrogenase activity in onion root cells.[Pubmed: 24241824]J Chem Ecol. 1994 Oct;20(10):2471-80.
|
| Animal Research | Effects of phytic acid and xanthotoxin on growth and detoxification in caterpillars.[Pubmed: 11545369]Tritrophic effects of xanthotoxin on the polyembryonic parasitoid Copidosoma sosares (Hymenoptera: Encyrtidae).[Pubmed: 18523826]J Chem Ecol. 2008 Jun;34(6):783-90.Plant chemistry can have deleterious effects on insect parasitoids, which include the reduction in body size, increased development time, and increased mortality. We examined the effects of Xanthotoxin, a linear furanocoumarin, on the polyembryonic encyrtid wasp Copidosoma sosares, a specialist parasitoid that attacks the parsnip webworm, Depressaria pastinacella, itself a specialist on furanocoumarin-producing plants. Furanocoumarins, allelochemicals abundant in the Apiaceae and Rutaceae, are toxic to a wide range of herbivores.
J Chem Ecol. 2001 Sep;27(9):1763-73.Phytic acid is abundant in the fruits and seeds of many plants and is found in foliage to a lesser extent. Among its several properties, phytic acid is a potent chelator of essential minerals and proteins; thus, the possibility exists that heme-based enzymes such as cytochrome P450 monooxygenases in herbivores are detrimentally affected by phytic acid via chelation of dietary iron.
|

Xanthotoxin Dilution Calculator

Xanthotoxin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.6253 mL | 23.1267 mL | 46.2535 mL | 92.5069 mL | 115.6337 mL |
| 5 mM | 0.9251 mL | 4.6253 mL | 9.2507 mL | 18.5014 mL | 23.1267 mL |
| 10 mM | 0.4625 mL | 2.3127 mL | 4.6253 mL | 9.2507 mL | 11.5634 mL |
| 50 mM | 0.0925 mL | 0.4625 mL | 0.9251 mL | 1.8501 mL | 2.3127 mL |
| 100 mM | 0.0463 mL | 0.2313 mL | 0.4625 mL | 0.9251 mL | 1.1563 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Threo-methylphenidate hydrochloride
Catalog No.:BCC5818
CAS No.:298-59-9
- Carbamazepine
Catalog No.:BCC4378
CAS No.:298-46-4
- Silydianin
Catalog No.:BCN2388
CAS No.:29782-68-1
- Nudifloside C
Catalog No.:BCN7491
CAS No.:297740-99-9
- Nudifloside B
Catalog No.:BCN7474
CAS No.:297740-98-8
- Teniposide
Catalog No.:BCC3864
CAS No.:29767-20-2
- Altenuene
Catalog No.:BCN7392
CAS No.:29752-43-0
- Altretamine hydrochloride
Catalog No.:BCC4114
CAS No.:2975-00-0
- Loganetin
Catalog No.:BCN5202
CAS No.:29748-10-5
- Luteolin-7-O-glucuronide
Catalog No.:BCN5338
CAS No.:29741-10-4
- Apigenin-7-glucuronide
Catalog No.:BCN5326
CAS No.:29741-09-1
- Dihydro-β-erythroidine hydrobromide
Catalog No.:BCC7341
CAS No.:29734-68-7
- Nitrotetrazolium Blue chloride
Catalog No.:BCC6465
CAS No.:298-83-9
- MTT
Catalog No.:BCC8031
CAS No.:298-93-1
- Desmethylbellidifolin
Catalog No.:BCN3868
CAS No.:2980-32-7
- H-Pro-NMe2
Catalog No.:BCC3019
CAS No.:29802-22-0
- Fatostatin A
Catalog No.:BCC6184
CAS No.:298197-04-3
- Shanzhiside
Catalog No.:BCN5203
CAS No.:29836-27-9
- Astilbin
Catalog No.:BCN5204
CAS No.:29838-67-3
- Pirenzepine dihydrochloride
Catalog No.:BCC6923
CAS No.:29868-97-1
- Ro 67-7476
Catalog No.:BCC6145
CAS No.:298690-60-5
- Amygdalin
Catalog No.:BCN5206
CAS No.:29883-15-6
- Astringin
Catalog No.:BCN3412
CAS No.:29884-49-9
- Sparteine sulfate pentahydrate
Catalog No.:BCN1267
CAS No.:299-39-8
Xanthotoxin prevents bone loss in ovariectomized mice through the inhibition of RANKL-induced osteoclastogenesis.[Pubmed:26809192]
Osteoporos Int. 2016 Jul;27(7):2335-2344.
UNLABELLED: Xanthotoxin (XAT) is extracted from the seeds of Ammi majus. Here, we reported that XAT has an inhibitory effect on osteoclastogenesis in vitro through the suppression of both receptor activator of nuclear factor-kappaB ligand (RANKL)-induced ROS generation and Ca(2+) oscillations. In vivo studies showed that XAT treatment decreases the osteoclast number, prevents bone loss, and restores bone strength in ovariectomized mice. INTRODUCTION: Excessive osteoclast formation and the resultant increase in bone resorption activity are key pathogenic factors of osteoporosis. In the present study, we have investigated the effects of XAT, a natural furanocoumarin, on the RANKL-mediated osteoclastogenesis in vitro and on ovariectomy-mediated bone loss in vivo. METHODS: Cytotoxicity of XAT was evaluated using bone marrow macrophages (BMMs). Osteoclast differentiation, formation, and fusion were assessed using the tartrate-resistant acid phosphatase (TRAP) stain, the actin cytoskeleton and focal adhesion (FAK) stain, and the fusion assay, respectively. Osteoclastic bone resorption was evaluated using the pit formation assay. Reactive oxygen species (ROS) generation and removal were evaluated using dichlorodihydrofluorescein diacetate (DCFH-DA). Ca(2+) oscillations and their downstream signaling targets were then detected. The ovariectomized (OVX) mouse model was adopted for our in vivo studies. RESULTS: In vitro assays revealed that XAT inhibited the differentiation, formation, fusion, and bone resorption activity of osteoclasts. The inhibitory effect of XAT on osteoclastogenesis was associated with decreased intracellular ROS generation. XAT treatment also suppressed RANKL-induced Ca(2+) oscillations and the activation of the resultant downstream calcium-CaMKK/PYK2 signaling. Through these two mechanisms, XAT downregulated the key osteoclastogenic factors nuclear factor of activated T cells c1 (NFATc1) and c-FOS. Our in vivo studies showed that XAT treatment decreases the osteoclast number, prevents bone loss, rescues bone microarchitecture, and restores bone strength in OVX mice. CONCLUSION: Our findings indicate that XAT is protective against ovariectomy-mediated bone loss through the inhibition of RANKL-mediated osteoclastogenesis. Therefore, XAT may be considered to be a new therapeutic candidate for treating osteoporosis.
Anticonvulsant effects of four linear furanocoumarins, bergapten, imperatorin, oxypeucedanin, and xanthotoxin, in the mouse maximal electroshock-induced seizure model: a comparative study.[Pubmed:21273683]
Pharmacol Rep. 2010 Nov-Dec;62(6):1231-6.
The aim of this study was to determine and compare the anticonvulsant activities of four natural furanocoumarins [bergapten (5-methoxypsoralen), imperatorin (8-isopentenyloxypsoralen), oxypeucedanin (5-epoxy-isopentenyloxypsoralen) and Xanthotoxin (8-methoxypsoralen)] in the maximal electroshock-induced seizure test in mice. The anticonvulsant effects of bergapten, imperatorin, oxypeucedanin, and Xanthotoxin were evaluated at 15, 30, 60 and 120 min after their systemic (intraperitoneal) administration. Tonic hind limb extension (seizure activity) was evoked in adult albino Swiss mice by a current (sine-wave, 25 mA, 500 V, 50 Hz, 0.2 s stimulus duration) delivered via auricular electrodes. The time courses of protection by bergapten, imperatorin, oxypeucedanin and Xanthotoxin against maximal electroshock-induced seizures revealed that 300 mg/kg imperatorin and Xanthotoxin (C-8 substituted derivatives of psoralen) exerted strong anticonvulsant activity, whereas 300 mg/kg bergapten and oxypeucedanin (C-5 substituted derivatives of psoralen) did not produce any anticonvulsant activity in this model. In conclusion, imperatorin and Xanthotoxin protected the animals against maximal electroshock-induced seizures, whereas bergapten and oxypeucedanin, despite their chemical and structural similarities to Xanthotoxin and imperatorin, exerted no anticonvulsant activity in this seizure test.
Effect of coumarin and xanthotoxin on mitochondrial structure, oxygen uptake, and succinate dehydrogenase activity in onion root cells.[Pubmed:24241824]
J Chem Ecol. 1994 Oct;20(10):2471-80.
At concentrations in which they occur on the plant surface and retard mitosis, coumarin and Xanthotoxin lowered uptake of oxygen (by 60 and 30%, respectively) by meristematic cells ofAllium cepa root tips. They caused changes in the structure of the mitochondrial matrix to become dense, and protrusions of mitochondrial membranes were visible parallelling their hypertrophy, indicating alteration in the structure and physiology of these organelles. Coumarin and, to a lesser extent, Xanthotoxin increased succinate dehydrogenase production in mitochondria and also in the cytoplasm, indicating changes in membrane permeability. Changes in oxygen uptake and mitochondrial structure, in addition to the retardation of mitosis, may be the reason these compounds act as allelochemicals after they have been removed from the plant surface and reach the root meristem.
Effects of methoxsalen, a CYP2A5/6 inhibitor, on nicotine dependence behaviors in mice.[Pubmed:24859605]
Neuropharmacology. 2014 Oct;85:67-72.
Metabolism of nicotine to inactive cotinine by hepatic enzyme CYP2A6 is the principal pathway by which active nicotine is removed from circulation. We therefore hypothesized that inhibition of mouse CYP2A5, the ortolog of human CYP2A6, by methoxsalen (8-methoxypsoralen) alter dependence-related behaviors of nicotine in the mouse. Conditioned place preference (CPP) test was used to assess the appetitive reward-like properties and precipitated nicotine withdrawal to assess physical (somatic and hyperalgesia) and affective (anxiety-related behaviors) measures. The nicotine plasma levels were also measured with or without methoxsalen pretreatment. Methoxsalen (15 and 30 mg/kg, intraperitoneally) pretreatment enhanced nicotine-induced preference in mice (p<0.05). However, there was a lack of enhancement of nicotine in the CPP test after the highest dose of the CYP-2A5 inhibitor. Similarly to the CPP results, repeated administration of methoxsalen increased the intensity of mecamylamine-precipitated withdrawal signs. The potentiation of nicotine preference and withdrawal intensity by methoxsalen was accompanied by significant increase in nicotine plasma levels in mice (p<0.05). Finally, methoxsalen enhanced the ability of a very low dose of nicotine (0.05 mg/kg) to reverse withdrawal signs in mice undergoing spontaneous withdrawal after chronic nicotine infusion (p<0.05). In conclusion, inhibition of nicotine metabolism by methoxsalen alters the behavioral effects of nicotine in the mouse. Combining CYP2A6 inhibitors with low dose nicotine replacement therapies may have a beneficial role in smoking cessation because it will decrease the drug elimination rate and maintain plasma and brain nicotine levels.
Tritrophic effects of xanthotoxin on the polyembryonic parasitoid Copidosoma sosares (Hymenoptera: Encyrtidae).[Pubmed:18523826]
J Chem Ecol. 2008 Jun;34(6):783-90.
Plant chemistry can have deleterious effects on insect parasitoids, which include the reduction in body size, increased development time, and increased mortality. We examined the effects of Xanthotoxin, a linear furanocoumarin, on the polyembryonic encyrtid wasp Copidosoma sosares, a specialist parasitoid that attacks the parsnip webworm, Depressaria pastinacella, itself a specialist on furanocoumarin-producing plants. Furanocoumarins, allelochemicals abundant in the Apiaceae and Rutaceae, are toxic to a wide range of herbivores. In this study, we reared parasitized webworms on artificial diets containing no Xanthotoxin (control) or low or high concentrations of Xanthotoxin. Clutch sizes of both male and female C. sosares broods were more than 20% smaller when they developed in hosts fed the diet containing high concentrations of Xanthotoxin. Xanthotoxin concentration in the artificial diet had no effect on the development time of C. sosares, nor did it have an effect on the body size (length of hind tibia) of individual adult male and female C. sosares in single-sex broods. Webworms fed artificial diets containing low or high concentrations of Xanthotoxin were not significantly smaller, and their development time was similar to that of webworms fed a Xanthotoxin-free diet. Mortality of webworms was not affected by Xanthotoxin in their artificial diet. Therefore, dietary Xanthotoxin did not appear to affect C. sosares via impairment of host health. However, unmetabolized Xanthotoxin was found in D. pastinacella hemolymph where C. sosares embryos develop. Hemolymph concentrations were fourfold greater in webworms fed the high-Xanthotoxin-containing diet than in webworms fed the low-Xanthotoxin-containing diet. We failed to detect any Xanthotoxin metabolism by either C. sosares embryos or precocious larvae. Therefore, the observed tritrophic effects of Xanthotoxin are likely to be due to the effects of Xanthotoxin after direct contact in the hemolymph rather than to the effects of compromised host quality.
Effects of phytic acid and xanthotoxin on growth and detoxification in caterpillars.[Pubmed:11545369]
J Chem Ecol. 2001 Sep;27(9):1763-73.
Phytic acid is abundant in the fruits and seeds of many plants and is found in foliage to a lesser extent. Among its several properties, phytic acid is a potent chelator of essential minerals and proteins; thus, the possibility exists that heme-based enzymes such as cytochrome P450 monooxygenases in herbivores are detrimentally affected by phytic acid via chelation of dietary iron. Mortality, growth performance, and P450-mediated metabolism of Xanthotoxin, a plant allelochemical, were examined in the presence of phytic acid in three lepidopteran species: a polyphagous seed-feeding species (Heliothis virescens), a polyphagous foliage-feeding species (Trichoplusia ni), and a species oligophagous on immature reproductive structures of two genera of Apiaceae (Depressaria pastinacella). While first instar H. virescens experienced no increase in mortality after 120 hours on a diet containing 1% phytic acid compared to a control diet, both T. ni and D. pastinacella experienced virtually complete mortality over the same time period. Ultimate instars of all three species experienced reductions in relative growth rates (RGR) and relative consumption rates (RCR) in the presence of phytic acid, although the only species to experience reduced digestive efficiency (ECI) was H. virescens. Cytochrome P450-mediated metabolism of Xanthotoxin was reduced 60% in the presence of phytic acid in D. pastinacella, although metabolism remained unaffected in the two noctuids. These studies suggest a defensive function of phytic acid in addition to its primary functions of phosphorus storage, energy storage, and cell wall precursor source.


