CI 976Selective ACAT inhibitor CAS# 114289-47-3 |
- Hydroxyfasudil
Catalog No.:BCC1635
CAS No.:105628-72-6
- Y-27632 dihydrochloride
Catalog No.:BCC1273
CAS No.:129830-38-2
- Hydroxyfasudil hydrochloride
Catalog No.:BCC1636
CAS No.:155558-32-0
- H-1152
Catalog No.:BCC1615
CAS No.:451462-58-1
- H-1152 dihydrochloride
Catalog No.:BCC1616
CAS No.:871543-07-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 114289-47-3 | SDF | Download SDF |
| PubChem ID | 122327 | Appearance | Powder |
| Formula | C23H39NO4 | M.Wt | 393.57 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 50 mM in ethanol and to 100 mM in DMSO | ||
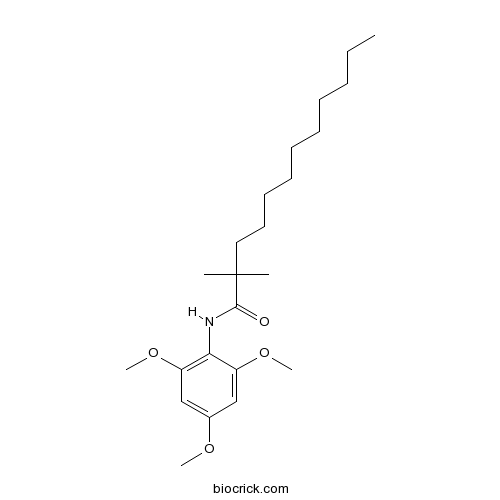
| Chemical Name | 2,2-dimethyl-N-(2,4,6-trimethoxyphenyl)dodecanamide | ||
| SMILES | CCCCCCCCCCC(C)(C)C(=O)NC1=C(C=C(C=C1OC)OC)OC | ||
| Standard InChIKey | WAFNZAURAWBNDZ-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C23H39NO4/c1-7-8-9-10-11-12-13-14-15-23(2,3)22(25)24-21-19(27-5)16-18(26-4)17-20(21)28-6/h16-17H,7-15H2,1-6H3,(H,24,25) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective acyl-coenzyme A:cholesterol acyltransferase (ACAT) inhibitor (IC50 = 0.073 μM). Lowers non-high density lipoprotein (HDL)-cholesterol and increases HDL-cholesterol concentrations in rats with pre-established dyslipidemia. Orally active. Also inhibits Golgi-associated LPL acyltransferase (LPAT) activity (IC50 = 15 μM). |

CI 976 Dilution Calculator

CI 976 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5408 mL | 12.7042 mL | 25.4084 mL | 50.8169 mL | 63.5211 mL |
| 5 mM | 0.5082 mL | 2.5408 mL | 5.0817 mL | 10.1634 mL | 12.7042 mL |
| 10 mM | 0.2541 mL | 1.2704 mL | 2.5408 mL | 5.0817 mL | 6.3521 mL |
| 50 mM | 0.0508 mL | 0.2541 mL | 0.5082 mL | 1.0163 mL | 1.2704 mL |
| 100 mM | 0.0254 mL | 0.127 mL | 0.2541 mL | 0.5082 mL | 0.6352 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Puerarin 6''-O-xyloside
Catalog No.:BCN2780
CAS No.:114240-18-5
- PAOPA
Catalog No.:BCC6353
CAS No.:114200-31-6
- Z-Ala-OH
Catalog No.:BCC3055
CAS No.:1142-20-7
- Butyraxanthone B
Catalog No.:BCN3603
CAS No.:1141754-81-5
- Kuguacin N
Catalog No.:BCN3056
CAS No.:1141453-73-7
- Kuguacin J
Catalog No.:BCN3055
CAS No.:1141453-65-7
- Tirandalydigin
Catalog No.:BCN1860
CAS No.:114118-91-1
- Cabozantinib malate (XL184)
Catalog No.:BCC4388
CAS No.:1140909-48-3
- Ibandronic acid
Catalog No.:BCC5204
CAS No.:114084-78-5
- Humantenidine
Catalog No.:BCN4754
CAS No.:114027-39-3
- 16-Epivoacarpine
Catalog No.:BCN3940
CAS No.:114027-38-2
- Phaclofen
Catalog No.:BCC6562
CAS No.:114012-12-3
- Soyacerebroside I
Catalog No.:BCN6022
CAS No.:114297-20-0
- Guanosine Hydrate
Catalog No.:BCC5326
CAS No.:1143525-19-2
- AZD5363
Catalog No.:BCC1073
CAS No.:1143532-39-1
- TAK 21d
Catalog No.:BCC5609
CAS No.:1143578-94-2
- Fmoc-D-Leu-OH
Catalog No.:BCC3511
CAS No.:114360-54-2
- PF-8380
Catalog No.:BCC1857
CAS No.:1144035-53-9
- WYE-125132 (WYE-132)
Catalog No.:BCC4608
CAS No.:1144068-46-1
- Beta-Furoyleupatolide
Catalog No.:BCN6407
CAS No.:114437-24-0
- BNP (1-32), human
Catalog No.:BCC1039
CAS No.:114471-18-0
- Oroxin B
Catalog No.:BCN1203
CAS No.:114482-86-9
- Z-Ser-OH
Catalog No.:BCC2743
CAS No.:1145-80-8
- (-)-U-50488 hydrochloride
Catalog No.:BCC6666
CAS No.:114528-79-9
The lysophospholipid acyltransferase antagonist CI-976 inhibits a late step in COPII vesicle budding.[Pubmed:18331383]
Traffic. 2008 May;9(5):786-97.
The mechanism of coat protein (COP)II vesicle fission from the endoplasmic reticulum (ER) remains unclear. Lysophospholipid acyltransferases (LPATs) catalyze the conversion of various lysophospholipids to phospholipids, a process that can promote spontaneous changes in membrane curvature. Here, we show that 2,2-methyl-N-(2,4,6,-trimethoxyphenyl)dodecanamide (CI-976), a potent LPAT inhibitor, reversibly inhibited export from the ER in vivo and the formation of COPII vesicles in vitro. Moreover, CI-976 caused the rapid and reversible accumulation of cargo at ER exit sites (ERESs) containing the COPII coat components Sec23/24 and Sec13/31 and a marked enhancement of Sar1p-mediated tubule formation from ERESs, suggesting that CI-976 inhibits the fission of assembled COPII budding elements. These results identify a small molecule inhibitor of a very late step in COPII vesicle formation, consistent with fission inhibition, and demonstrate that this step is likely facilitated by an ER-associated LPAT.
Iron loaded ferritin secretion and inhibition by CI-976 in Aedes aegypti larval cells.[Pubmed:19168145]
Comp Biochem Physiol B Biochem Mol Biol. 2009 Apr;152(4):352-63.
Ferritin is a multimer of 24 subunits of heavy and light chains. In mammals, iron taken into cells is stored in ferritin or incorporated into iron-containing proteins. Very little ferritin is found circulating in mammalian serum; most is retained in the cytoplasm. Female mosquitoes, such as Aedes aegypti (yellow fever mosquito, Diptera), require a blood meal for oogenesis. Mosquitoes receive a potentially toxic level of iron in the blood meal which must be processed and stored. We demonstrate by (59)Fe pulse-chase experiments that cultured A. aegypti larval CCL-125 cells take up iron from culture media and store it in ferritin found mainly in the membrane fraction and secrete iron-loaded ferritin. We observe that in these larval cells ferritin co-localizes with ceramide-containing membranes in the absence of iron. With iron treatment, ferritin is found associated with ceramide-containing membranes as well as in cytoplasmic non-ceramide vesicles. Treatment of CCL-125 cells with iron and CI-976, an inhibitor of lysophospholipid acyl transferases, disrupts ferritin secretion with a concomitant decrease in cell viability. Interfering with ferritin secretion may limit the ability of mosquitoes to adjust to the high iron load of the blood meal and decrease iron delivery to the ovaries reducing egg numbers.
A unique lysophospholipid acyltransferase (LPAT) antagonist, CI-976, affects secretory and endocytic membrane trafficking pathways.[Pubmed:15972316]
J Cell Sci. 2005 Jul 15;118(Pt 14):3061-71.
Previous studies have shown that inhibition of a Golgi-complex-associated lysophospholipid acyltransferase (LPAT) activity by the drug CI-976 stimulates Golgi tubule formation and subsequent redistribution of resident Golgi proteins to the endoplasmic reticulum (ER). Here, we show that CI-976 stimulates tubule formation from all subcompartments of the Golgi complex, and often these tubules formed independently, i.e. individual tubules usually did not contain markers from different subcompartments. Whereas the cis, medial and trans Golgi membranes redistributed to the ER, the trans Golgi network (TGN) collapsed back to a compact juxtanuclear position similar to that seen with brefeldin A (BFA) treatment. Also similar to BFA, CI-976 induced the formation of endosome tubules, but unlike BFA, these tubules did not fuse with TGN tubules. Finally, CI-976 produced an apparently irreversible block in the endocytic recycling pathway of transferrin (Tf) and Tf receptors (TfRs) but had no direct effect on Tf uptake from the cell surface. Tf and TfRs accumulated in centrally located, Rab11-positive vesicles indicating that CI-976 inhibits export of cargo from the central endocytic recycling compartment. These results, together with previous studies, demonstrate that CI-976 inhibits multiple membrane trafficking steps, including ones found in the endocytic and secretory pathways, and imply a wider role for lysophospholipid acyltransferases in membrane trafficking.
A lysophospholipid acyltransferase antagonist, CI-976, creates novel membrane tubules marked by intracellular phospholipase A1 KIAA0725p.[Pubmed:23378048]
Mol Cell Biochem. 2013 Apr;376(1-2):151-61.
CI-976 is a lysophospholipid acyltransferase antagonist that is known to affect secretory and endocytic membrane-trafficking pathways likely by increasing the lysophospholipid content in membranes. Our previous study suggested that lysophospholipids formed through the action of an intracellular phospholipase A(1), KIAA0725p (also known as DDHD2 and iPLA(1)gamma), may be important for the association of this enzyme with membranes. In this study, we examined the effect of CI-976 on the membrane association of KIAA0725p. While in HeLa cells KIAA0725p is localized in the Golgi and cytosol, in mouse embryonic fibroblasts (MEFs), it was found to be principally localized in the cytosol with some on post-endoplasmic reticulum compartments including the cis-Golgi. Treatment of MEFs with CI-976 induced the redistribution of KIAA0725p to membrane tubules, which were in vicinity to fragmented mitochondria. These tubules were not decorated with canonical organelle markers including Golgi proteins. A human KIAA0725p mutant, which exhibits decreased membrane-binding ability, was also redistributed to membrane structures upon CI-976 treatment. Our data suggest that the association of KIAA0725p with membranes is regulated by lipid metabolism, and that CI-976 may create unique membrane structures that can be marked by KIAA0725p.
In vivo evidence that the lipid-regulating activity of the ACAT inhibitor CI-976 in rats is due to inhibition of both intestinal and liver ACAT.[Pubmed:8429262]
J Lipid Res. 1993 Feb;34(2):279-94.
CI-976, a new trimethoxy fatty acid anilide, is a potent and specific inhibitor of liver and intestinal acyl coenzyme A:cholesterol acyltransferase (ACAT) in vitro. Several in vivo approaches were used to determine the efficacy and sites of action of this compound in rats. CI-976 decreased non-high density lipoprotein (HDL)-cholesterol and increased HDL-cholesterol in rats with pre-established dyslipidemia. High performance gel chromatographic separation of plasma lipoproteins also revealed that CI-976, but not CL 277,082, lowered low density lipoprotein (LDL)-cholesterol and elevated HDL-cholesterol. Bay o 2752, octimibate, melinamide, and SaH 58-035 were all less potent in vivo compared to CI-976 and CL 277,082, and CI-976 produced the greatest decrease in liver cholesteryl esters. Subcutaneous (SC) administration of CI-976 was also efficacious in cholesterol-fed animals. In sucrose-fed rats, oral and SC CI-976 administration potently lowered plasma triglycerides. Hepatic cholesteryl ester accumulation in the ethinyl estradiol-treated rat was also diminished by orally administered CI-976. ACAT activity and cholesteryl ester mass were dose-dependently decreased in the livers from cholesterol-fed rats treated with CI-976, suggesting a direct effect on the liver. In both hypercholesterolemic and hypertriglyceridemic models, CI-976 also decreased plasma apoB concentrations. In other experiments radiolabeled CI-976 accumulated in the liver after multiple doses. Time-dependent changes in biliary lipid and bile acid secretion suggested that free cholesterol did not accumulate in the liver but instead was excreted as such or as bile acid. Finally, inhibition of endogenous and exogenous intestinal cholesterol absorption was demonstrated using several in vivo techniques. The combined data strongly supports the hypothesis that orally administered CI-976 inhibits both intestinal and hepatic ACAT, and that both of these enzymes may be determinants of plasma lipid concentrations in the rat.


