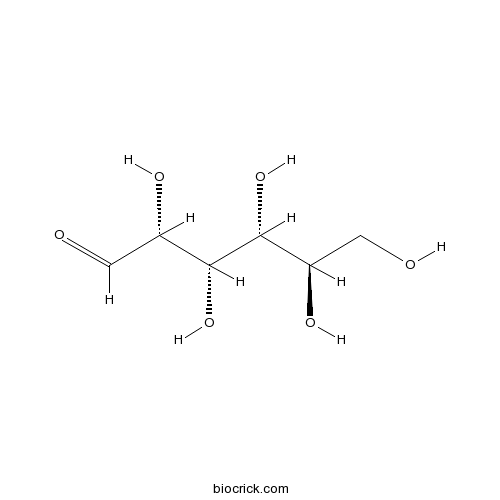
D-(+)-GlucoseNaturally occurring monosaccharide CAS# 50-99-7 |
- Gatifloxacin
Catalog No.:BCC1064
CAS No.:112811-59-3
- Doxorubicin (Adriamycin) HCl
Catalog No.:BCC1117
CAS No.:25316-40-9
- Etoposide
Catalog No.:BCC1151
CAS No.:33419-42-0
- Genistein
Catalog No.:BCN5499
CAS No.:446-72-0
- Ellagic acid
Catalog No.:BCN5533
CAS No.:476-66-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 50-99-7 | SDF | Download SDF |
| PubChem ID | 107526 | Appearance | Powder |
| Formula | C6H12O6 | M.Wt | 180.16 |
| Type of Compound | Miscellaneous | Storage | Desiccate at -20°C |
| Synonyms | D-Glucose; Grape sugar; Glucopyranose;aldehydo-D-glucose | ||
| Solubility | DMSO : 50 mg/mL (277.53 mM; Need ultrasonic) H2O : ≥ 50 mg/mL (277.53 mM) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | (2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanal | ||
| SMILES | C(C(C(C(C(C=O)O)O)O)O)O | ||
| Standard InChIKey | GZCGUPFRVQAUEE-SLPGGIOYSA-N | ||
| Standard InChI | InChI=1S/C6H12O6/c7-1-3(9)5(11)6(12)4(10)2-8/h1,3-6,8-12H,2H2/t3-,4+,5+,6+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Dextrose, a simple sugar (monosaccharide), is an important carbohydrate in biology, it exhibits marked antibacterial activity against Staphylococcus aureus,Escherichia coli and Pyocyanine. D-(+)-Glucose can prevent MPP+ toxicity, attenuate the loss of ATP, but do not reverse the complete inhibition of mitochondrial O2 consumption (MOC). |
| Targets | Antifection | ATP |
| In vivo | d-(+)-Glucose rescue against 1-methyl-4-phenylpyridinium toxicity through anaerobic glycolysis in neuroblastoma cells[Reference: WebLink]Brain Research Volume 962, Issues 1–2, 7 February 2003, Pages 48–60The active neurotoxin of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), 1-methyl-4-phenylpyridinium (MPP+), exerts its lethal effect by inhibiting Complex I of the electron transport chain (ETC). MPP+ shuts down aerobic oxidative phosphorylation and ETC-mediated ATP synthesis.
|
| Structure Identification | Tetrahedron,2000,11(23): 4701-8.Synthesis of novel diphosphines from d-(+)-glucose. Use in asymmetric hydrogenation[Reference: WebLink]A new family of bidentate diphosphine ligands which contain a glucofuranoside as a simple but highly effective chiral backbone are reported. These ligands are prepared in a few steps from readily available D-(+)-Glucose.
Academic Forum of Nan Du, 2001.Synthesis and Antibacterial Activity of Cu(Ⅱ) Complexes With D-(+)-Glucose Thiosemicarbazone[Reference: WebLink]
|

D-(+)-Glucose Dilution Calculator

D-(+)-Glucose Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.5506 mL | 27.7531 mL | 55.5062 mL | 111.0124 mL | 138.7655 mL |
| 5 mM | 1.1101 mL | 5.5506 mL | 11.1012 mL | 22.2025 mL | 27.7531 mL |
| 10 mM | 0.5551 mL | 2.7753 mL | 5.5506 mL | 11.1012 mL | 13.8766 mL |
| 50 mM | 0.111 mL | 0.5551 mL | 1.1101 mL | 2.2202 mL | 2.7753 mL |
| 100 mM | 0.0555 mL | 0.2775 mL | 0.5551 mL | 1.1101 mL | 1.3877 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Dextrose is the biologically active form of Glucose (D-Glucose).
- Ephedrine Hydrochloride
Catalog No.:BCC8322
CAS No.:50-98-6
- Floxuridine
Catalog No.:BCC1187
CAS No.:50-91-9
- Thymidine
Catalog No.:BCN5622
CAS No.:50-89-5
- Ascorbic acid
Catalog No.:BCN2207
CAS No.:50-81-7
- Aspirin (Acetylsalicylic acid)
Catalog No.:BCC2097
CAS No.:50-78-2
- Actinomycin D
Catalog No.:BCC2385
CAS No.:50-76-0
- 1,3:2,4-Di-p-methylbenyliedene sorbitol
Catalog No.:BCC4847
CAS No.:54686-97-4
- 5-Hydroxytryptamine
Catalog No.:BCC9204
CAS No.:50-67-9
- Niclosamide
Catalog No.:BCC5081
CAS No.:50-65-7
- Chloroquine diphosphate
Catalog No.:BCC3915
CAS No.:50-63-5
- Oxytocin
Catalog No.:BCC5435
CAS No.:50-56-6
- Reserpine
Catalog No.:BCN4960
CAS No.:50-55-5
- Nordihydroguaiaretic acid
Catalog No.:BCC1805
CAS No.:500-38-9
- L-Mimosine
Catalog No.:BCC5450
CAS No.:500-44-7
- Apoatropine
Catalog No.:BCN1869
CAS No.:500-55-0
- Convolamine
Catalog No.:BCN1905
CAS No.:500-56-1
- Yangonin
Catalog No.:BCN3565
CAS No.:500-62-9
- Kawain
Catalog No.:BCN3564
CAS No.:500-64-1
- Rhapontigenin
Catalog No.:BCN3515
CAS No.:500-65-2
- Olivetol
Catalog No.:BCN4629
CAS No.:500-66-3
- 3,5-Dimethoxyphenol
Catalog No.:BCN7198
CAS No.:500-99-2
- Rilpivirine
Catalog No.:BCC1897
CAS No.:500287-72-9
- GANT61
Catalog No.:BCC1090
CAS No.:500579-04-4
- Securinol A
Catalog No.:BCN6987
CAS No.:5008-48-0


