Dimethyl phthalateCAS# 131-11-3 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 131-11-3 | SDF | Download SDF |
| PubChem ID | 8554 | Appearance | Oil |
| Formula | C10H10O4 | M.Wt | 194.2 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | dimethyl benzene-1,2-dicarboxylate | ||
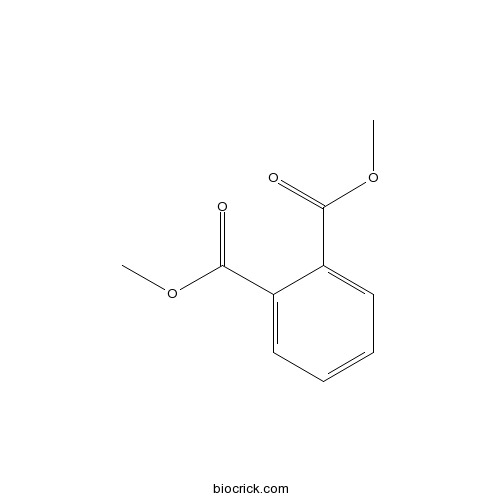
| SMILES | COC(=O)C1=CC=CC=C1C(=O)OC | ||
| Standard InChIKey | NIQCNGHVCWTJSM-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C10H10O4/c1-13-9(11)7-5-3-4-6-8(7)10(12)14-2/h3-6H,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Dimethyl phthalate is an organic compound, used as an insect repellent for mosquitoes and flies. |
| In vitro | Degradation of dimethyl phthalate in solutions and soil slurries by persulfate at ambient temperature.[Pubmed: 24637446]J Hazard Mater. 2014 Apr 30;271:202-9.The degradation of Dimethyl phthalate (DMP) by persulfate at ambient temperature (T=20-40°C) was investigated in aqueous solutions and soil slurries to assess the feasibility of using persulfate to remediate Dimethyl phthalate contaminated soil and groundwater. Laboratory evaluation of dimethyl phthalate treated wristbands against three predominant mosquito (Diptera: Culicidae) vectors of disease.[Pubmed: 20556923]Eur Rev Med Pharmacol Sci. 2010 May;14(5):443-8.
Kinetic study of the removal of dimethyl phthalate from an aqueous solution using an anion exchange resin.[Pubmed: 24510529]Environ Sci Pollut Res Int. 2014 May;21(10):6571-7.Phthalate acid esters are becoming an important class of pollutants in wastewaters. This study addresses the kinetics of removal of Dimethyl phthalate (DMP) using the anion exchange resin D201-OH from an aqueous solution. |
| Kinase Assay | Potential toxicity of phthalic acid esters plasticizer: interaction of dimethyl phthalate with trypsin in vitro.[Pubmed: 25496445 ]J Agric Food Chem. 2015 Jan 14;63(1):75-84.Dimethyl phthalate (DMP) is widely used as a plasticizer in industrial processes and has been reported to possess potential toxicity to the human body. |

Dimethyl phthalate Dilution Calculator

Dimethyl phthalate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.1493 mL | 25.7467 mL | 51.4933 mL | 102.9866 mL | 128.7333 mL |
| 5 mM | 1.0299 mL | 5.1493 mL | 10.2987 mL | 20.5973 mL | 25.7467 mL |
| 10 mM | 0.5149 mL | 2.5747 mL | 5.1493 mL | 10.2987 mL | 12.8733 mL |
| 50 mM | 0.103 mL | 0.5149 mL | 1.0299 mL | 2.0597 mL | 2.5747 mL |
| 100 mM | 0.0515 mL | 0.2575 mL | 0.5149 mL | 1.0299 mL | 1.2873 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- alpha-Yohimbine
Catalog No.:BCN6166
CAS No.:131-03-3
- VU 0360172 hydrochloride
Catalog No.:BCC6141
CAS No.:1309976-62-2
- 15-Methoxymkapwanin
Catalog No.:BCN6498
CAS No.:1309920-99-7
- MRS 4062 triethylammonium salt
Catalog No.:BCC6134
CAS No.:1309871-50-8
- Sephin1
Catalog No.:BCC3980
CAS No.:13098-73-2
- (±)-CPSI 1306
Catalog No.:BCC6161
CAS No.:1309793-47-2
- H 89 2HCl
Catalog No.:BCC4997
CAS No.:130964-39-5
- Marumoside A
Catalog No.:BCN7702
CAS No.:1309604-34-9
- Wittifuran X
Catalog No.:BCN4794
CAS No.:1309478-07-6
- K145
Catalog No.:BCC4305
CAS No.:1309444-75-4
- GPR40 Activator 1
Catalog No.:BCC4125
CAS No.:1309435-60-6
- Cerberic acid B
Catalog No.:BCN4715
CAS No.:1309362-77-3
- Pimpinellin
Catalog No.:BCN6168
CAS No.:131-12-4
- N-Acetylneuraminic acid
Catalog No.:BCN2204
CAS No.:131-48-6
- Oxybenzone
Catalog No.:BCC5445
CAS No.:131-57-7
- Meptyldinocap
Catalog No.:BCC5468
CAS No.:131-72-6
- 8-Epiloganic acid
Catalog No.:BCC8956
CAS No.:82509-41-9
- Boldenone undecylenate
Catalog No.:BCC8896
CAS No.:13103-34-9
- GNF179 Metabolite
Catalog No.:BCC5176
CAS No.:1310455-86-7
- NB-598
Catalog No.:BCC1786
CAS No.:131060-14-5
- Tubastatin A HCl
Catalog No.:BCC3877
CAS No.:1310693-92-5
- NVP-BGJ398 phosphate
Catalog No.:BCC1814
CAS No.:1310746-10-1
- Miltipolone
Catalog No.:BCN3222
CAS No.:131086-61-8
- 5,7-Dichlorokynurenic acid
Catalog No.:BCC6592
CAS No.:131123-76-7
Kinetic study of the removal of dimethyl phthalate from an aqueous solution using an anion exchange resin.[Pubmed:24510529]
Environ Sci Pollut Res Int. 2014 May;21(10):6571-7.
Phthalate acid esters are becoming an important class of pollutants in wastewaters. This study addresses the kinetics of removal of Dimethyl phthalate (DMP) using the anion exchange resin D201-OH from an aqueous solution. The effects of various factors on the removal rate and efficiency were investigated. An overall initial removal rate (OIRR) law and a pseudo first-order kinetic (PFOK) model were also developed. The internal diffusion of DMP within the resin phase of D201-OH is the rate-controlling step. Optimization of the particle size and pore structure of the resin D201-OH, the DMP concentration, and the reaction temperature can improve the DMP removal rate. The hydrolysis reaction of DMP catalyzed by D201-OH indicates an overall reaction order of 1.76, a value that is between the first order and the second order. The apparent activation energy of the reaction is 34.6 kJ/mol, which is below the homogeneous alkaline hydrolysis activation energy of 44.3 kJ/mol. The OIRR law can quantify the initial removal rate under different conditions. The results also show that the theoretical DMP removal efficiency predicted by the PFOK model agrees well with the experimentally determined values. Our research provides valuable insights into the primary parameters influencing the kinetic process, which enables a focused improvement in the removal or hydrolysis rate for similar processes.
Potential toxicity of phthalic acid esters plasticizer: interaction of dimethyl phthalate with trypsin in vitro.[Pubmed:25496445]
J Agric Food Chem. 2015 Jan 14;63(1):75-84.
Dimethyl phthalate (DMP) is widely used as a plasticizer in industrial processes and has been reported to possess potential toxicity to the human body. In this study, the interaction between DMP and trypsin in vitro was investigated. The results of fluorescence, UV-vis, circular dichroism, and Fourier transform infrared spectra along with cyclic voltammetric measurements indicated that the remarkable fluorescence quenching and conformational changes of trypsin resulted from the formation of a DMP-trypsin complex, which was driven mainly by hydrophobic interactions. The molecular docking and trypsin activity assay showed that DMP primarily interacted with the catalytic triad of trypsin and led to the inhibition of trypsin activity. The dimensions of the individual trypsin molecules were found to become larger after binding with DMP by atomic force microscopy imaging. This study offers a comprehensive picture of DMP-trypsin interaction, which is expected to provide insights into the toxicological effect of DMP.
Laboratory evaluation of dimethyl phthalate treated wristbands against three predominant mosquito (Diptera: Culicidae) vectors of disease.[Pubmed:20556923]
Eur Rev Med Pharmacol Sci. 2010 May;14(5):443-8.
UNLABELLED: OBJECTIVES AND MATERIALS AND METHODS: The repellent efficacy of Dimethyl phthalate (DMP) treated wristband was determined against mosquitoes, viz Anopheles stephensi Liston, Aedes aegypti Linnaeus, Culex quinquefasciatus Say at two concentrations viz., 1.5 and 2.0 mg/cm2 under the laboratory conditions. DMP treated wristband had shown variable degrees of repellency impact against different mosquito species. RESULTS: Its offered higher reduction of man landing rate against Anopheles stephensi at both concentrations of 1.5 (81.1%) and 2.0 mg/cm2 (87.0%). 79.8% and 84.8% of protection achieved against Culex quinquefasciatus at concentrations of 1.5 and 2.0 mg/cm2, respectively. 74.4 and 86.5% of reduction of man landing rates were obtained against Aedes aegypti at concentrations of 1.5 and 2.0 mg/cm2 respectively. The reduction of man-landing rate evaluations were confirmed by t-test compared between control group and each experimental group. The t-test result shows at 1.5 (p = 0.0026; t = 19.2; df = 2) as well 2.0 mg/cm2 (p = 0.0025; t = 19.8; df = 2) are extremely significant to reduce the man vector contact. CONCLUSION: The present data suggest that DMP treated wristbands are most promising against both day and night-biting mosquitoes and significantly reducing the man-vector contact. Therefore, it could serve as a potential as means of personal protection device against insect nuisance biting and insect-borne disease when and where other kinds of personal protection measures are impossible and impracticable.
Degradation of dimethyl phthalate in solutions and soil slurries by persulfate at ambient temperature.[Pubmed:24637446]
J Hazard Mater. 2014 Apr 30;271:202-9.
The degradation of Dimethyl phthalate (DMP) by persulfate at ambient temperature (T=20-40 degrees C) was investigated in aqueous solutions and soil slurries to assess the feasibility of using persulfate to remediate DMP contaminated soil and groundwater. First, the effects of temperature, initial oxidant concentration, initial DMP concentration and initial solution pH on the removal of DMP and TOC were studied in aqueous solutions. The results show that persulfate at 40 degrees C can effectively mineralize DMP. Furthermore, dimethyl 4-hydroxyl phthalate, maleic acid and oxalic acid were identified as the degradation intermediates, and degradation pathways were proposed. Lastly, persulfate at 40 degrees C was applied to remediate soil spiked with DMP at approximately 600 mg/kg. The results show that persulfate at 40 degrees C is highly effective for the remediation of DMP contaminated soil. Overall, this study provides fundamental and practical knowledge for the treatment of emerging phthalate esters (PAEs) contaminated soil and groundwater, as well as PAEs contaminated industrial wastewater, with persulfate at ambient temperature.


