Flavopiridol hydrochlorideCDK inhibitor, potent and selective CAS# 131740-09-5 |
- LEE011
Catalog No.:BCC3926
CAS No.:1211441-98-3
- THZ1
Catalog No.:BCC4005
CAS No.:1604810-83-4
- Roscovitine (Seliciclib,CYC202)
Catalog No.:BCC1105
CAS No.:186692-46-6
- BMS265246
Catalog No.:BCC3741
CAS No.:582315-72-8
- AZD-5438
Catalog No.:BCC3689
CAS No.:602306-29-6
- R547
Catalog No.:BCC3927
CAS No.:741713-40-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 131740-09-5 | SDF | Download SDF |
| PubChem ID | 9910986 | Appearance | Powder |
| Formula | C21H21Cl2NO5 | M.Wt | 438.3 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | L 86-8275 | ||
| Solubility | H2O : ≥ 20 mg/mL (45.63 mM) DMF : 7.69 mg/mL (17.55 mM; Need ultrasonic) *"≥" means soluble, but saturation unknown. | ||
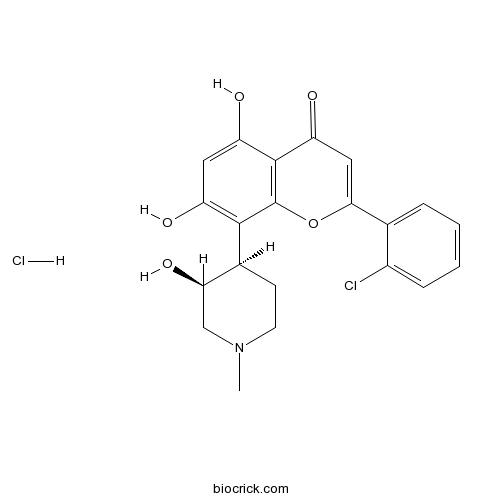
| Chemical Name | 2-(2-Chlorophenyl)-5,7-dihydroxy-8- | ||
| SMILES | [Cl-].CN1CC[C@@H]([C@H](O)C1)c2c(O)cc(O)c3C(=O)C=C(Oc23)c4ccccc4Cl.[H+] | ||
| Standard InChIKey | LGMSNQNWOCSPIK-LWHGMNCYSA-N | ||
| Standard InChI | InChI=1S/C21H20ClNO5.ClH/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22;/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3;1H/t12-,17+;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Cyclin-dependent kinase (CDK) inhibitor that causes cell cycle arrest at G1 and G2 phase. Potently inhibits the growth of breast and lung cancer cell lines (IC50 = 25 - 160 nM) in vitro. |

Flavopiridol hydrochloride Dilution Calculator

Flavopiridol hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2815 mL | 11.4077 mL | 22.8154 mL | 45.6308 mL | 57.0386 mL |
| 5 mM | 0.4563 mL | 2.2815 mL | 4.5631 mL | 9.1262 mL | 11.4077 mL |
| 10 mM | 0.2282 mL | 1.1408 mL | 2.2815 mL | 4.5631 mL | 5.7039 mL |
| 50 mM | 0.0456 mL | 0.2282 mL | 0.4563 mL | 0.9126 mL | 1.1408 mL |
| 100 mM | 0.0228 mL | 0.1141 mL | 0.2282 mL | 0.4563 mL | 0.5704 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Flavopiridol hydrochloride is a selective inhibitor of CDK1, CDK2, CDK4 and CDK6 with IC50 value all ~41 nM as well as CDK7 with 300nM [1] [2] [3].
CDKs are protein kinase families which involve in the process of regulating transcription, mRNA processing, cell differentiation and cell cycle. It has been shown that CDKs are abnormally expressed in a variety of cells [3].
Flavopiridol hydrochloride is a potent CDK inhibitor and is different from UCN-01. When tested with breast carcinoma cell line MCF-7, flavopiridol hydrochloride treatment showed high inhibitory ability to arrest cell cycle at G1 phase through inhibiting CDK2 and CDK4 [1]. Treated seven lymphoma cell lines with flavopiridol hydrochloride at the concentration of 400 nM, it showed that flavopiridol hydrochloride induced cells apoptosis by inhibiting CDK4, CDK6, CDK7 or CDK9 which in turn inhibited Rb phosphorylation, anti-apoptotic preteins (Mcl-1 and XIAP) [4].
In mouse model with HL-60 subcutaneous xenograft, administration of flavopiridol hydrochloride (7.5 mg/kg) intravenously induced advanced stage animals complete regression with 91.67% and remained disease-free several months after one course of flavopiridol treatment [5].
It has also been reported that flavopiridol hydrochloride inhibited epidermal growth factoe receptor and protein kinase A with IC50 value of 21 and 122 μM, respectively [3].
References:
[1]. Carlson, B.A., et al., Flavopiridol induces G1 arrest with inhibition of cyclin-dependent kinase (CDK) 2 and CDK4 in human breast carcinoma cells. Cancer Res, 1996. 56(13): p. 2973-8.
[2]. Losiewicz, M.D., et al., Potent inhibition of CDC2 kinase activity by the flavonoid L86-8275. Biochem Biophys Res Commun, 1994. 201(2): p. 589-95.
[3]. Senderowicz, A.M., The cell cycle as a target for cancer therapy: basic and clinical findings with the small molecule inhibitors flavopiridol and UCN-01. Oncologist, 2002. 7 Suppl 3: p. 12-9.
[4]. Ema, Y., et al., Investigation of the cytotoxic effect of flavopiridol in canine lymphoma cell lines. Vet Comp Oncol, 2015.
[5]. Arguello, F., et al., Flavopiridol induces apoptosis of normal lymphoid cells, causes immunosuppression, and has potent antitumor activity In vivo against human leukemia and lymphoma xenografts. Blood, 1998. 91(7): p. 2482-90.
- Urolignoside
Catalog No.:BCN6758
CAS No.:131723-83-6
- Arbidol HCl
Catalog No.:BCC3722
CAS No.:131707-23-8
- (R)-(-)-4-Benzyl-3-propionyl-2-oxazolidinone
Catalog No.:BCC8392
CAS No.:131685-53-5
- Parsonsianine
Catalog No.:BCN2110
CAS No.:131683-36-8
- 8-(1-Chloro-2-hydroxy-3-methylbut-3-enyl)-7-methoxycoumarin
Catalog No.:BCN7058
CAS No.:131652-35-2
- Turmeronol A
Catalog No.:BCN6777
CAS No.:131651-37-1
- Chalepensin
Catalog No.:BCN7334
CAS No.:13164-03-9
- OPC 21268
Catalog No.:BCC7812
CAS No.:131631-89-5
- ACY-241
Catalog No.:BCC6460
CAS No.:1316215-12-9
- Rocilinostat (ACY-1215)
Catalog No.:BCC2144
CAS No.:1316214-52-4
- (+)-Pteryxin
Catalog No.:BCN3470
CAS No.:13161-75-6
- Amyloid Beta-peptide (25-35) (human)
Catalog No.:BCC1027
CAS No.:131602-53-4
- DAU 5884 hydrochloride
Catalog No.:BCC7263
CAS No.:131780-48-8
- Ugaxanthone
Catalog No.:BCN6775
CAS No.:13179-11-8
- Cardenolide B-1
Catalog No.:BCN4714
CAS No.:1318158-89-2
- Aphagranin A
Catalog No.:BCN6889
CAS No.:1318173-53-3
- (R,R)-2,6-Bis(4-isopropyl-2-oxazolin-2-yl)pyridine
Catalog No.:BCC8396
CAS No.:131864-67-0
- Lexacalcitol
Catalog No.:BCC1704
CAS No.:131875-08-6
- (3R)-(+)-1-Benzyl-3-(tert-butoxycarbonylamino)pyrrolidine
Catalog No.:BCC8389
CAS No.:131878-23-4
- 2-Hydroxyethyl Salicylate
Catalog No.:BCN3579
CAS No.:87-28-5
- Fudosteine
Catalog No.:BCC4661
CAS No.:13189-98-5
- Goitrin
Catalog No.:BCN2764
CAS No.:13190-34-6
- Solanesol
Catalog No.:BCN2596
CAS No.:13190-97-1
- Paricalcitol
Catalog No.:BCC1839
CAS No.:131918-61-1
Cross-talk between cell cycle induction and mitochondrial dysfunction during oxidative stress and nerve growth factor withdrawal in differentiated PC12 cells.[Pubmed:21557293]
J Neurosci Res. 2011 Aug;89(8):1302-15.
Neuronal death has been reported to involve mitochondrial dysfunction and cell cycle reentry. In this report, we used Nerve Growth Factor (NGF)-differentiated PC12 cells to investigate mechanisms linking mitochondrial dysfunction and cell cycle activation during neuronal death induced by NGF withdrawal and/or oxidative stress. We found that loss of survival following H(2) O(2) -induced oxidative stress or NGF deprivation was preceded by a decrease in mitochondrial membrane potential (DeltaPsim), increase in reactive oxygen species (ROS), and up-regulation of cyclin D1 and phosphorylation (Ser-780) of protein retinoblastoma (P-pRb), without an increase of proliferation rates. Treatment with H(2) O(2) , but not NGF deprivation, also induced the phosporylation (Ser-10) of p27(kip1) and the appearance of a cleaved P-p27(kip1) fragment of about 15 kDa. The extent of cell cycle activation appeared to be inversely correlated to the duration of toxic stimuli (pulse/continuous). H(2) O(2) -induced mitogenic responses appeared to be mediated by induction of P-MAPK and P-Akt and were blocked by p38MAPK and JNK inhibitors as well as by the CDK inhibitor flavopiridol (Flav) and by sodium selenite (Sel), a component of selenoproteins, including glutathione peroxidases. Inhibition of p38MAPK and JNK, instead, did not affect cyclin D1 changes following NGF deprivation. Finally, both Flav hydrochloride and Sel partially prevented mitochondrial dysfunction and cell death following NGF withdrawal or H(2) O(2) toxicity, but not during oxidative stress in the absence of NGF. Taken together, these data suggest that H(2) O(2) -induced oxidative stress can determine distinct patterns of mitogenic responses as a function of mitochondrial dysfunction depending on 1) intensity/duration of stress stimuli and/or 2) presence of NGF.
Kinetic analysis of the cooperation of P-glycoprotein (P-gp/Abcb1) and breast cancer resistance protein (Bcrp/Abcg2) in limiting the brain and testis penetration of erlotinib, flavopiridol, and mitoxantrone.[Pubmed:20304939]
J Pharmacol Exp Ther. 2010 Jun;333(3):788-96.
A synergistic effect of P-glycoprotein (P-gp)/Abcb1a and breast cancer resistance protein (Bcrp)/Abcg2 was reported to limit the brain penetration of their common substrates. This study investigated this based on pharmacokinetics using Mdr1a/1b(-/-), Bcrp(-/-), and Mdr1a/1b(-/-)/Bcrp(-/-) mice. Comparison of the brain- and testis-to-plasma ratios (C(brain)/C(plasma) and C(testis)/C(plasma), respectively) of the reference compounds quinidine and dantrolene for P-gp and Bcrp, respectively, indicates that impairment of either P-gp and Bcrp did not cause any change in the efflux activities of Bcrp or P-gp, respectively, at both the blood-brain barrier (BBB) and blood-testis barrier (BTB). The C(brain)/C(plasma) and C(testis)/C(plasma) of the common substrates erlotinib, flavopiridol, and mitoxantrone were markedly increased in Mdr1a/1b(-/-)/Bcrp(-/-) mice even compared with Mdr1a/1b(-/-) and Bcrp(-/-) mice. Efflux activities by P-gp and Bcrp relative to passive diffusion at the BBB and BTB were separately evaluated based on the C(brain)/C(plasma) and C(testis)/C(plasma) in the knockout strains to the wild-type strain. P-gp made a larger contribution than Bcrp to the net efflux of the common substrates, but Bcrp activities were also significantly larger than passive diffusion. These parameters could reasonably account for the marked increase in C(brain)/C(plasma) and C(testis)/C(plasma) in the Mdr1a/1b(-/-)/Bcrp(-/-) mice. In conclusion, the synergistic effect of P-gp and Bcrp on C(brain)/C(plasma) and C(testis)/C(plasma) can be explained by their contribution to the net efflux at the BBB and BTB without any interaction between P-gp and Bcrp.
Smad3 deficiency protects mice from obesity-induced podocyte injury that precedes insulin resistance.[Pubmed:25945408]
Kidney Int. 2015 Aug;88(2):286-98.
Signaling by TGF-beta/Smad3 plays a key role in renal fibrosis. As obesity is one of the major risk factors of chronic and end-stage renal disease, we studied the role of Smad3 signaling in the pathogenesis of obesity-related renal disease. After switching to a high fat diet, the onset of Smad3 C-terminal phosphorylation, increase in albuminuria, and the early stages of peripheral and renal insulin resistance occurred at 1 day, and 4 and 8 weeks, respectively, in C57BL/6 mice. The loss of synaptopodin, a functional marker of podocytes, and phosphorylation of the Smad3 linker region (T179 and S213) appeared after 4 weeks of the high fat diet. This suggests a temporal pattern of Smad3 signaling activation leading to kidney injury and subsequent insulin resistance in the development of obesity-related renal disease. In vivo, Smad3 knockout attenuated the high fat diet-induced proteinuria, renal fibrosis, overall podocyte injury, and mitochondrial dysfunction in podocytes. In vitro palmitate caused a rapid activation of Smad3 in 30 min, loss of synaptopodin in 2 days, and impaired insulin signaling in 3 days in isolated mouse podocytes. Blockade of either Smad3 phosphorylation by SIS3 (a Smad3 inhibitor) or T179 phosphorylation by flavopiridol (a CDK9 inhibitor) prevented the palmitate-induced loss of synaptopodin and mitochondrial function in podocytes. Thus, Smad3 signaling plays essential roles in obesity-related renal disease and may be a novel therapeutic target.
Quantitative evaluation of the impact of active efflux by p-glycoprotein and breast cancer resistance protein at the blood-brain barrier on the predictability of the unbound concentrations of drugs in the brain using cerebrospinal fluid concentration as a surrogate.[Pubmed:21934030]
J Pharmacol Exp Ther. 2011 Dec;339(3):935-44.
This study investigated the impact of the active efflux mediated by P-glycoprotein (P-gp) and breast cancer resistance protein (Bcrp) at the blood-brain barrier (BBB) on the predictability of the unbound brain concentration (C(u,brain)) by the concentration in the cerebrospinal fluid (CSF) (C(u,CSF)) in rats. C(u,brain) is obtained as the product of the total brain concentration and unbound fraction in the brain (f(u,brain)) determined in vitro in brain slices. Twenty-five compounds, including P-gp and/or Bcrp substrates, were given a constant intravenous infusion, and their plasma, brain, and CSF concentrations were determined. P-gp and/or Bcrp substrates, such as verapamil, loperamide, flavopiridol, genistein, quinidine, dantrolene, daidzein, cimetidine, and pefloxacin, showed a higher CSF-to-brain unbound concentration ratio (K(p,uu,CSF/brain)) compared with non-P-gp and non-Bcrp substrates. K(p,uu,CSF/brain) values of P-gp-specific (quinidine and verapamil) and Bcrp-specific (daidzein and genistein) substrates were significantly decreased in Mdr1a/1b(-/-) and Bcrp(-/-) mice, respectively. Furthermore, consistent with the contribution of P-gp and Bcrp to the net efflux at the BBB, K(p,uu,CSF/brain) values of the common substrates (flavopiridol and erlotinib) were markedly decreased in Mdr1a/1b(-/-)/Bcrp(-/-) mice, but only moderately or weakly in Mdr1a/1b(-/-) mice and negligibly in Bcrp(-/-) mice. In conclusion, predictability of C(u,brain) by C(u,CSF) decreases along with the net transport activities by P-gp and Bcrp at the BBB. C(u,CSF) of non-P-gp and non-Bcrp substrates can be a reliable surrogate of C(u,brain) for lipophilic compounds.
Gateways to clinical trials.[Pubmed:20664824]
Methods Find Exp Clin Pharmacol. 2010 Jun;32(5):331-88.
[(1)(1)C]RAC; (18)F-Fluoromisonidazole; 89-12; 9-[(1)(8)F]Fluoropropyl-(+)-dihydrotetrabenazine; Adalimumab, Adecatumumab, ADMVA, ADXS-11-001, Aflibercept, Agatolimod sodium, AGS-004, Alglucosidase alfa, Aliskiren fumarate, Alvocidib hydrochloride, AMG-108, AMG-853, Apixaban, Aripiprazole, Armodafinil, Atazanavir sulfate, Atomoxetine hydrochloride; Bevacizumab, BioMatrix Flex drug eluting stent, Biphasic insulin aspart, Bortezomib, Bosentan; Caspofungin acetate, Cediranib, Cetuximab, ChimeriVax-Dengue, Choriogonadotropin alfa, Cinacalcet hydrochloride, Cizolirtine citrate, Clofarabine, Cocaine conjugate vaccine, CX-717; Darbepoetin alfa, Dasatinib, Decitabine, Denosumab, Desvenlafaxine succinate, Dexamethasone sodium phosphate, Dienogest, Diphencyprone, Doripenem, DTaP-HepB-IPV, Dutasteride; E-7010, Ecallantide, Ecstasy, Eicosapentaenoic acid/docosahexaenoic acid, Emtricitabine, Enfuvirtide, Erlotinib hydrochloride, Eszopiclone, Etonogestrel/ethinyl estradiol, Etoricoxib, Everolimus, Everolimus-eluting coronary stent EVT-201, Ezetimibe, Ezetimibe/simvastatin; Ferumoxytol, Fesoterodine fumavate, Figitumumab, Filgrastim, Fingolimod hydrochloride, Fluticasone furoate, Fluval P, Fluzone, Fondaparinux sodium, Fulvestrant, Fungichromin; Gamma-hydroxybutyrate sodium, Gefitinib, GHB-01L1, GLY-230, GSK-1349572; Hib-MenCY-TT, Hib-TT, HPV-6/11/16/18, Hydrocodone bitartrate; IC-51, Icatibant acetate, Imatinib mesylate, Immunoglobulin intravenous (human), Indetanib, Influenza A (H1N1) 2009 Monovalent Vaccine, Inhalable human insulin, Insulin glargine, Insulin glulisine, Interferon-beta, Ispinesib mesylate, Ixabepilone; Laromustine, Latanoprost/timolol maleate, L-Citrulline, Lenalidomide, Lexatumumab, Linezolid, Lopinavir/ritonavir, Lutropin alfa; Mapatumumab, MDX-066, MDX-1388, Mepolizumab, Methoxy polyethylene glycol-epoetin-beta, Metreleptin, Micafungin sodium, Mometasone furoate/oxymetazoline hydrochloride, Mx-dnG1, Mycophenolic acid sodium salt; Nabiximols, Natalizumab, Nemonoxacin, Norelgestromin/ethinyl estradiol; Oblimersen sodium, Ocriplasmin, Olmesartan medoxomil, Omacetaxine mepesuccinate; Paclitaxel-eluting stent, Pagoclone, Paliperidone, Panitumumab, Pazopanib hydrochloride, PCV7, Pegaptanib octasodium, Peginterferon alfa-2a, Peginterferon alfa-2b/ ribavirin, Pegvisomant, Pemetrexed disodium, Perifosine, Pimecrolimus, Pitavastatin calcium, Plerixafor hydrochloride, Plitidepsin, Posaconazole, Pregabalin, Progesterone capriate; Raltegravir potassium, Ramucirumab, Ranelic acid distrontium salt, Rasburicase, Recombinant Bet V1, Recombinant human insulin, rhFSH, Rolofylline, Romidepsin, Romiplostim, Rosuvastatin calcium; Sapacitabine, Sevelamer carbonate, Sinecatechins, Sirolimus-eluting stent, Sitagliptin phosphate monohydrate, SN-29244, Sorafenib, Sugammadex sodium, Sunitinib malate; Tadalafil, Tafenoquine, Talnetant, Tanezumab, Tapentadol hydrochloride, Tasocitinib citrate, Technosphere/Insulin, Telcagepant, Tenofovir disoproxil fumarate, Teriparatide, Ticagrelor, Tigecycline, Tiotropium bromide, Tipifarnib, Tocilizumab, TS-041; Ulipristal acetate, Urtoxazumab, Ustekinumab; Vandetanib, Varenicline tartrate, Vicriviroc, Voriconazole, Vorinostat, VRC-HIVADV014-00-VP, VRC-HIVDNA016-00-VP; Zoledronic acid monohydrate.
The cyclin-dependent kinase inhibitor flavopiridol potentiates the effects of topoisomerase I poisons by suppressing Rad51 expression in a p53-dependent manner.[Pubmed:18381438]
Cancer Res. 2008 Apr 1;68(7):2312-20.
The results of a phase I clinical trial of the topoisomerase I (Topo I) poison CPT-11 followed by the cyclin-dependent kinase inhibitor flavopiridol in patients with advanced solid tumors indicate that patients whose tumors were wild-type, but not mutant, for p53 obtained the most clinical benefit from this combination therapy. We elected to elucidate the mechanistic basis for this effect in isogenic-paired HCT116 colon cancer cells that were either wild-type (+/+) or null (-/-) for p53. With the combination therapy of SN-38 (the active metabolite of CPT-11) followed by flavopiridol, the induction of apoptosis was 5-fold greater in the p53+/+ cells compared with the p53-/- cells. This sequential treatment induced phosphorylation of p53 at Ser(15), which interacted with Rad51, a DNA repair protein involved in homologous recombination. Rad51 bound to p53-Ser(15) within the first 5 hours of combination therapy, and then was transcriptionally suppressed at 24 hours by flavopiridol only in p53+/+ cells. Microarray analysis also revealed suppression of Rad51 in a p53-dependent manner. Depletion of Rad51 by small interfering RNA (siRNA) sensitized both p53+/+ and p53-/- cells to SN-38-induced apoptosis with increase of gamma H2AX, a marker of DNA damage. Conversely, overexpression of Rad51 rescued p53+/+ cells from SN-->F-induced apoptosis. Because flavopiridol inhibits Cdk9, we found that inhibition of Cdk9 by DRB or by siRNA could recapitulate the flavopiridol effects, with suppression of Rad51 and induction of apoptosis only in p53+/+ cells. In conclusion, after DNA damage by Topo I poisons, flavopiridol targets homologous recombination through a p53-dependent down-regulation of Rad51, resulting in enhancement of apoptosis.
Growth inhibition with reversible cell cycle arrest of carcinoma cells by flavone L86-8275.[Pubmed:1279187]
J Natl Cancer Inst. 1992 Nov 18;84(22):1736-40.
BACKGROUND: Previous studies have shown that polyhydroxylated flavonoids such as quercetin and genistein can inhibit tumor cell growth in vitro, and preliminary in vivo studies of the flavone L86-8275 have shown growth inhibition of LX529 and A549 lung carcinomas. L86-8275 [(-)cis-5,7-dihydroxy-2-(2-chlorophenyl)-8[4-(3-hydroxy-1-methyl)- piperidinyl]-4H-1-benzopyran-4-one] is a flavone of novel structure. PURPOSE: The purpose of this study was to determine in vitro whether L86-8275 is a more potent inhibitor of growth in breast carcinoma and lung carcinoma cells than quercetin or genistein. METHODS: We studied the effects of L86-8275 on cell growth in seven breast carcinoma cell lines and five lung carcinoma cell lines. MDA468 breast carcinoma was then selected for further study. Cell proliferation was measured by a colorimetric dye reduction assay; synthesis of DNA, RNA, and protein by incorporation of the radioactive metabolic precursors thymidine, uridine, or leucine, respectively; adenosine triphosphate (ATP) content by a luciferase-mediated bioluminescence reaction; and cell cycle progression by the use of cell-synchronizing drugs (aphidicolin and nocodazole) and flow cytometry. RESULTS: L86-8275 was not cytotoxic to stationary-phase cells but reversibly inhibited the growth of cells in exponential growth phase. At concentrations of 25-160 nM, L86-8275 inhibited growth of human breast and lung carcinoma cell lines by 50%. MDA468 breast carcinoma cells were 60-fold and 400-fold more sensitive to L86-8275 than to quercetin and genistein, respectively. By 24 hours after addition of L86-8275, DNA synthesis in MDA468 cells was inhibited by greater than 95%, protein synthesis by 80%, and RNA synthesis by 40%-60%, under conditions that preserved cellular ATP levels at approximately 80%-90% of control values. When MDA468 cells released from aphidicolin-induced cell cycle arrest were exposed to 200 nM L86-8275, they completed the S phase but arrested in G2. When cells released from nocodazole-induced cell cycle arrest were exposed to 200 nM L86-8275, they completed mitosis but arrested in G1. CONCLUSIONS: L86-8275 is a potent, yet reversible, growth-inhibitory flavone that can selectively block cell cycle progression in vitro at more than one point in the cell cycle. IMPLICATIONS: These findings suggest that L86-8275 is a candidate for further preclinical development, as well as a model for the synthesis of other flavonoids that might potently delay cell cycle progression to achieve inhibition of tumor growth. Future studies need to address optimal schedules for antiproliferative activity in vivo and inhibition of clonogenic activity.


