HU 211NMDA antagonist, novel and non-competitive CAS# 112924-45-5 |
- WZ4003
Catalog No.:BCC4363
CAS No.:1214265-58-3
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 112924-45-5 | SDF | Download SDF |
| PubChem ID | 107778 | Appearance | Powder |
| Formula | C25H38O3 | M.Wt | 386.57 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Dexanabinol | ||
| Solubility | Soluble to 100 mM in DMSO and to 100 mM in ethanol | ||
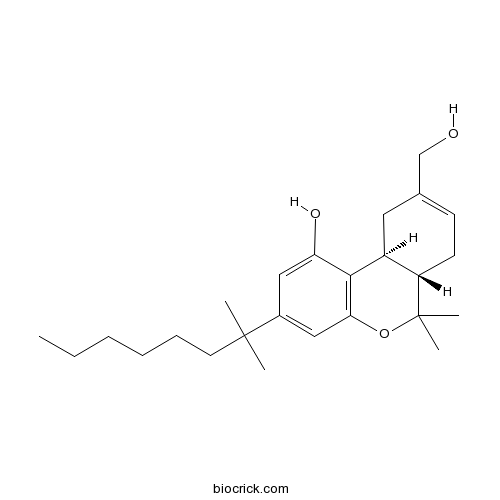
| Chemical Name | (6aS,10aS)-9-(hydroxymethyl)-6,6-dimethyl-3-(2-methyloctan-2-yl)-6a,7,10,10a-tetrahydrobenzo[c]chromen-1-ol | ||
| SMILES | CCCCCCC(C)(C)C1=CC2=C(C3CC(=CCC3C(O2)(C)C)CO)C(=C1)O | ||
| Standard InChIKey | SSQJFGMEZBFMNV-PMACEKPBSA-N | ||
| Standard InChI | InChI=1S/C25H38O3/c1-6-7-8-9-12-24(2,3)18-14-21(27)23-19-13-17(16-26)10-11-20(19)25(4,5)28-22(23)15-18/h10,14-15,19-20,26-27H,6-9,11-13,16H2,1-5H3/t19-,20-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | NMDA antagonist (IC50 = 11 μM for inhibition of [3H]MK-801 binding to rat forebrain membranes). Protects against NMDA- and quisqualate-induced neurotoxicity (EC50 = 3.8 μM) and enhances dopamine D1 receptor activity. Inhibits NF-κB, reducing TNF-α, IL-6 and nitric oxide production, and acts as a free radical scavenger. Exhibits beneficial effects in experimental models of multiple sclerosis, bacterial meningitis, septic shock, epilepsy, and traumatic and ischemic brain injury. Brain penetrant. |

HU 211 Dilution Calculator

HU 211 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5869 mL | 12.9343 mL | 25.8685 mL | 51.7371 mL | 64.6713 mL |
| 5 mM | 0.5174 mL | 2.5869 mL | 5.1737 mL | 10.3474 mL | 12.9343 mL |
| 10 mM | 0.2587 mL | 1.2934 mL | 2.5869 mL | 5.1737 mL | 6.4671 mL |
| 50 mM | 0.0517 mL | 0.2587 mL | 0.5174 mL | 1.0347 mL | 1.2934 mL |
| 100 mM | 0.0259 mL | 0.1293 mL | 0.2587 mL | 0.5174 mL | 0.6467 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
HU 211 is a novel and non-competitive antagonist of NMDA [1].
N-Methyl-D-aspartate (NMDA) is an amino acid derivative that acts as a specific agonist at the NMDA receptor and only binds to and regulates the NMDA receptor.
HU 211 is a novel and non-competitive NMDA antagonist. In culture neurones, HU 211 inhibited NMDA-mediated neurotoxicity in a dose dependent way with EC50 value of 3.8 µM. Also, HU 211 inhibited [3H]MK-801 binding to rat forebrain membranes in a competitive way with Ki value of 11.0 µM [1]. In rat alveolar macrophage cell line and murine peritoneal macrophages, HU 211 inhibited nitric oxide and TNFα production induced by lipopolysaccharide (LPS) [4].
In a global ischemia gerbil model, HU-211 (4 mg/kg) significantly induced neuroprotection in the CA1 subfield of the hippocampus [2]. In the rat with closed head injury (CHI), HU-211 (5 mg/kg) significantly improved neurological severity score (NSS) and slightly reduced edema. In the Morris water maze, HU-211 significantly improved the abilities impaired by CHI [3]. In BALB/c mice injected with 10 mg/kg LPS, HU-211 reduced lethality to 9 and 67%. In Sprague-Dawley rats, HU-211 abolished the hypotensive response induced by LPS [4].
References:
[1]. Eshhar N, Striem S, Biegon A. HU-211, a non-psychotropic cannabinoid, rescues cortical neurones from excitatory amino acid toxicity in culture. Neuroreport, 1993, 5(3): 237-240.
[2]. Bar-Joseph A, Berkovitch Y, Adamchik J, et al. Neuroprotective activity of HU-211, a novel NMDA antagonist, in global ischemia in gerbils. Mol Chem Neuropathol, 1994, 23(2-3): 125-135.
[3]. Shohami E, Novikov M, Bass R. Long-term effect of HU-211, a novel non-competitive NMDA antagonist, on motor and memory functions after closed head injury in the rat. Brain Res, 1995, 674(1): 55-62.
[4]. Gallily R, Yamin A, Waksmann Y, et al. Protection against septic shock and suppression of tumor necrosis factor alpha and nitric oxide production by dexanabinol (HU-211), a nonpsychotropic cannabinoid. J Pharmacol Exp Ther, 1997, 283(2): 918-924.
- Boc-D-Asp(OcHex)-OH
Catalog No.:BCC3372
CAS No.:112898-18-7
- BzATP triethylammonium salt
Catalog No.:BCC7643
CAS No.:112898-15-4
- Raltitrexed
Catalog No.:BCC4457
CAS No.:112887-68-0
- Mosapride Citrate
Catalog No.:BCC1065
CAS No.:112885-42-4
- Mosapride
Catalog No.:BCC4078
CAS No.:112885-41-3
- Fmoc-2-Nal-OH
Catalog No.:BCC3292
CAS No.:112883-43-9
- Fmoc-N-Me-Nle-OH
Catalog No.:BCC3299
CAS No.:112883-42-8
- Fmoc-D-Nle-OH
Catalog No.:BCC3300
CAS No.:112883-41-7
- Fmoc-D-Met-OH
Catalog No.:BCC3532
CAS No.:112883-40-6
- (24S)-MC 976
Catalog No.:BCC1291
CAS No.:112849-14-6
- (24R)-MC 976
Catalog No.:BCC1289
CAS No.:112828-09-8
- 24R-Calcipotriol
Catalog No.:BCC1304
CAS No.:112827-99-3
- PD128907 HCl
Catalog No.:BCC4469
CAS No.:112960-16-4
- Calcipotriol
Catalog No.:BCC1444
CAS No.:112965-21-6
- Tokinolide B
Catalog No.:BCN2753
CAS No.:112966-16-2
- IM-12
Catalog No.:BCC5487
CAS No.:1129669-05-1
- Estradiol diproppionate
Catalog No.:BCC8960
CAS No.:113-38-2
- Chlorprothixene
Catalog No.:BCC3753
CAS No.:113-59-7
- Chlorpheniramine Maleate
Catalog No.:BCC4526
CAS No.:113-92-8
- Potassium benzylpenicillin
Catalog No.:BCC9126
CAS No.:113-98-4
- VU 0155069
Catalog No.:BCC7715
CAS No.:1130067-06-9
- 7-O-Methylrosmanol
Catalog No.:BCN7276
CAS No.:113085-62-4
- 3-Deoxysappanone B
Catalog No.:BCN6012
CAS No.:113122-54-6
- (S)-(+)-Niguldipine hydrochloride
Catalog No.:BCC6947
CAS No.:113145-69-0
Dexanabinol: dexanabinone, HU 211, PA 50211, PRS 211007, sinnabidol.[Pubmed:12757406]
Drugs R D. 2003;4(3):185-7.
Dexanabinol [HU 211, dexanabinone, sinnabidol, PA 50211, PRS 211007] is a synthetic, non-psychotropic tetrahydro-cannabinoid. Dexanabinol lacks cannabimimetic activity, and is a functional antagonist of the NMDA receptor with antioxidant and anti-tumour necrosis factor-alpha properties. Dexanabinol is in clinical trials for traumatic brain injury (head injuries), glaucoma and mild cognitive impairment, and is being investigated preclinically for its potential in the treatment of multiple sclerosis. Pharmos has a licensing agreement with the Hebrew University, Israel, and is seeking a partner for development and commercialisation of the dexanabinol family of compounds. The Financial Times (ft.com) reported in March 2001 that the market for brain trauma could be worth approximately 500 million US dollars, according to estimates by Pharmos. The company was said to have 26 million US dollars in capital at the time, most of which would be used taking dexanabinol through regulatory submission, according to the Financial Times.
Antidepressant effects of curcumin and HU-211 coencapsulated solid lipid nanoparticles against corticosterone-induced cellular and animal models of major depression.[Pubmed:27757031]
Int J Nanomedicine. 2016 Oct 3;11:4975-4990.
Major depression is a complex neuropsychiatric disorder with few treatment approaches. The use of nontargeted antidepressants induced many side effects with their low efficacy. A more precise targeting strategy is to develop nanotechnology-based drug delivery systems; hence, we employed solid lipid nanoparticles (SLNs) to encapsulate HU-211 and curcumin (Cur). The antidepressant effects of the dual-drug nanoparticles (Cur/SLNs-HU-211) for major depression treatment were investigated in corticosterone-induced cellular and animal models of major depression. Cur/SLNs-HU-211 can effectively protect PC12 cells from corticosterone-induced apoptosis and can release more dopamine, which may be associated with the higher uptake of Cur/SLNs-HU-211 shown by cellular uptake behavior analysis. Additionally, Cur/SLNs-HU-211 significantly reduced the immobility time in forced swim test, enhanced fall latency in rotarod test, and improved the level of dopamine in mice blood. Cur/SLNs-HU-211 can deliver more Cur to the brain and thus produce a significant increase in neurotransmitters level in brain tissue, especially in the hippocampus and striatum. The results of Western blot and immunofluorescence revealed that Cur/SLNs-HU-211 can significantly enhance the expression of CB1, p-MEK1, and p-ERK1/2. Our study suggests that Cur/SLNs-HU-211 may have great potential for major depression treatment.
The protective effect of dexanabinol (HU-211) on nitric oxide and cysteine protease-mediated neuronal death in focal cerebral ischemia.[Pubmed:18404379]
Neurochem Res. 2008 Sep;33(9):1683-91.
We hypothesized that dexanabinol can prevent neuronal death by protecting neuronal lysosomes from nitric oxide (NO)-mediated toxicity, and in turn, by suppressing the release of cathepsins during cerebral ischemia. Focal cerebral ischemia was induced in two sets of animals by permanent middle cerebral artery occlusion. The first set was used to monitor NO concentration and cathepsin activity, while the second was used for histological examination with hematoxylin and eosin, and TUNEL staining. In post-ischemic brain tissue, NO content and cathepsin B and L activity increased (p < 0.05). Dexanabinol treatment reduced NO concentration and cathepsin activity to the control level (p > 0.05). The number of eosinophilic and apoptotic neurons increased in the post-ischemic cerebral cortex (p < 0.05). However, dexanabinol treatment lowered both of these (p < 0.05). We conclude that dexanabinol might be a useful agent for the treatment of stroke patients.
Synthesis, radio-synthesis and in vitro evaluation of terminally fluorinated derivatives of HU-210 and HU-211 as novel candidate PET tracers.[Pubmed:28210722]
Org Biomol Chem. 2017 Mar 1;15(9):2086-2096.
We report the synthesis of terminally fluorinated HU-210 and HU-211 analogues (HU-210F and HU-211F, respectively) and their biological evaluation as ligands of cannabinoid receptors (CB1 and CB2) and N-methyl d-aspartate receptor (NMDAR). [(18)F]-labelled HU-210F was radiosynthesised from the bromo-substituted precursor. In vitro assays showed that both HU-210F and HU-211F retain the potent pharmacological profile of HU-210 and HU-211, suggesting that [(18)F]-radiolabelled HU-210F and HU-211F could have potential as PET tracers for in vivo imaging.
The cannabinoid dexanabinol is an inhibitor of the nuclear factor-kappa B (NF-kappa B).[Pubmed:15380375]
Neuropharmacology. 2004 Sep;47(4):580-92.
Exogenous and endogenous cannabinoids have been shown to have neuroprotective effects in vitro and in vivo. Although many of the pharmacological effects of cannabinoids have been identified, the mechanism of neuroprotection still represents a controversy. Here we demonstrate for the first time protective effects of the synthetic cannabinoid dexanabinol by inhibiting apoptosis in a neuron-like cell line using nuclear staining and FACS analysis and in primary neurons. We provide further evidence of inhibition of nuclear factor-kappakappa B (NF-kappaB) by dexanabinol: Dexanabinol inhibits (1) phosphorylation and degradation of the inhibitor of NF-kappaB IkappaBalpha and translocation of NF-kappaB to the nucleus; dexanabinol reduces (2) the transcriptional activity of NF-kappaB and (3) mRNA accumulation of the NF-kappaB target genes tumor necrosis factor-alpha and interleukin-6 (TNF-alpha and IL-6). Dexanabinol does not bind to cannabinoid (CB) receptors 1 and 2. To investigate the mechanism of action, we employed the non-antioxidant CB1 receptor agonist WIN 55,212-2 and the antioxidant cannabinol, which binds to CB1 receptors only weakly. Both cannabinoids mimicked the effect of dexanabinol on NF-kappaB and apoptosis. This suggests that neither the antioxidant properties of cannabinoids nor binding to CB1 or CB2 receptors are responsible for the inhibition of NF-kappaB activity and apoptosis. Our results clearly demonstrate that dexanabinol inhibits NF-kappaB. NF-kappaB has been shown to be involved in brain damage and to promote neuronal cell death in vitro and in in vivo models of ischemic and neurodegenerative neurological diseases.
Protection against septic shock and suppression of tumor necrosis factor alpha and nitric oxide production by dexanabinol (HU-211), a nonpsychotropic cannabinoid.[Pubmed:9353414]
J Pharmacol Exp Ther. 1997 Nov;283(2):918-24.
Dexanabinol, HU-211, a synthetic cannabinoid devoid of psychotropic effects, improves neurological outcome in models of brain trauma, ischemia and meningitis. Recently, HU-211 was found to inhibit brain tumor necrosis factor (TNFalpha) production after head injury. In the present study, we demonstrate the ability of HU-211 to suppress TNFalpha production and to rescue mice and rats from endotoxic shock after LPS (Escherichia coli 055:B5) inoculation. In BALB/c mice, a dose of 10 mg/kg LPS, injected i.p., caused 57% and 100% mortality, at 24 and 48 hr, respectively. HU-211, administered i.p. 30 min before lipopolysaccharide (LPS), reduced lethality to 9 and 67% at these time points (P < .05). When coinjected with D-galactoseamine (i.p.), LPS was 100% lethal within 24 hr, whereas eight hourly injections of HU-211 caused mortality of C57BL/6 mice to drop to 10% (P < .001). Administration of LPS to Sprague-Dawley rats resulted in a 30% reduction in the mean arterial blood pressure within 30 min, which persisted for 3 hr. HU-211, given 2 to 3 min before LPS, completely abolished the typical hypotensive response. Furthermore, the drug also markedly suppressed in vitro TNFalpha production and nitric oxide generation (by >90%) by both murine peritoneal macrophages and rat alveolar macrophage cell line exposed to LPS. HU-211 may, therefore, have therapeutic implications in the treatment of TNFalpha-mediated pathologies.
Interaction of dexanabinol (HU-211), a novel NMDA receptor antagonist, with the dopaminergic system.[Pubmed:9424014]
Eur J Pharmacol. 1997 Nov 12;338(3):205-13.
The interaction of 7-hydroxy-delta6-tetrahydrocannabinol 1,1-dimethylheptyl (Dexanabinol: HU-211), a novel NMDA receptor antagonist, with the dopaminergic system was examined using in vitro and in vivo systems. HU-211 (50 or 100 microM) inhibited the binding of [3H]R(+)-8-chloro-2,3,4,5-tetrahydro-3-methyl-5-phenyl-1H-3-benzazepi n-7-ol hydrochloride ([3H]SCH-23390), a dopamine D1 receptor antagonist, by 29.7 +/- 1.8% and 52.7 +/- 6.3%, respectively. HU-211 10 microM, like the dopamine D1 receptor agonist R(+)-1-phenyl-2,3,4,5-tetrahydro-(1H)-3-benzazepine-7,8-diol hydrochloride (SKF-38393), enhanced the conversion of [3H]adenine to cyclic AMP (cAMP) (51.8 +/- 29.7% and 35.6 +/- 21.5% over control, respectively). The HU-211-induced increase was not inhibited by SCH-23390. HU-211 together with the dopamine D1 receptor agonist caused a synergistic elevation (314.7 +/- 14.3%). HU-211 reduced the catalepsy induced by dopamine receptor antagonists. At 10 mg/kg, HU-211 significantly (P < 0.001) reduced the catalepsy time induced by D1, D2 and non-selective dopamine receptor antagonists. Overall, the results of the present study demonstrate that HU-211 interacts with the dopaminergic system and enhances activity at the dopamine D1 receptor level. This activity may have implications in diseases involving the dopaminergic system, such as Parkinson's disease.
HU-211, a non-psychotropic cannabinoid, rescues cortical neurones from excitatory amino acid toxicity in culture.[Pubmed:8298080]
Neuroreport. 1993 Dec 13;5(3):237-40.
The present study examined potential neuroprotective effects of HU-211, a synthetic non-psychotropic cannabinoid with non-competitive NMDA antagonist properties on neurones exposed to various excitotoxins in culture. HU-211 was found to protect neurones from NMDA and quisqualate-induced toxicity but not that produced by AMPA or kainate. NMDA-mediated neurotoxicity was blocked by HU-211 in a dose dependent manner with an EC50 = 3.8 +/- 0.9 microM. Radioligand binding studies have shown that HU-211 inhibits the binding of [3H]MK-801 to rat forebrain membranes (KI = 11.0 microM +/- 1.323) in a competitive manner, but was unable to displace [3H]kainate and [3H]AMPA binding. These data suggest that the neuroprotective activity of HU-211 is directly associated with the NMDA receptor channel and possibly with the quisqualate receptor of the metabotropic class. Thus, HU-211 appears to act as an NMDA open channel blocker and shows promise as a novel neuroprotectant for clinical use.


