LersivirineNNRT inhibitor CAS# 473921-12-9 |
- Nevirapine
Catalog No.:BCC3820
CAS No.:129618-40-2
- Lamivudine
Catalog No.:BCC3801
CAS No.:134678-17-4
- Delavirdine
Catalog No.:BCC4300
CAS No.:136817-59-9
- Emtricitabine
Catalog No.:BCC3774
CAS No.:143491-57-0
- Tenofovir
Catalog No.:BCC2500
CAS No.:147127-20-6
- Delavirdine mesylate
Catalog No.:BCC4069
CAS No.:147221-93-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 473921-12-9 | SDF | Download SDF |
| PubChem ID | 16739244 | Appearance | Powder |
| Formula | C17H18N4O2 | M.Wt | 310.35 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | UK-453061 | ||
| Solubility | DMSO : 50 mg/mL (161.11 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
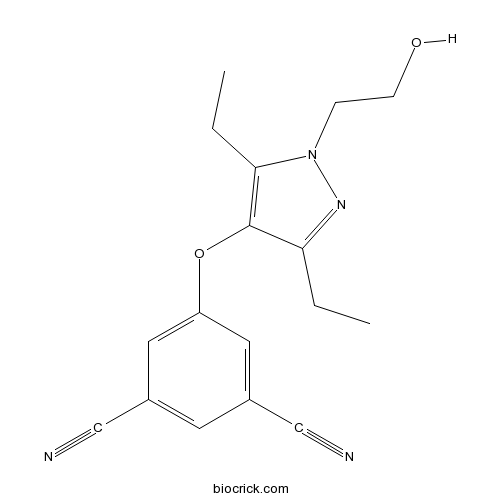
| Chemical Name | 5-[3,5-diethyl-1-(2-hydroxyethyl)pyrazol-4-yl]oxybenzene-1,3-dicarbonitrile | ||
| SMILES | CCC1=C(C(=NN1CCO)CC)OC2=CC(=CC(=C2)C#N)C#N | ||
| Standard InChIKey | MCPUZZJBAHRIPO-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H18N4O2/c1-3-15-17(16(4-2)21(20-15)5-6-22)23-14-8-12(10-18)7-13(9-14)11-19/h7-9,22H,3-6H2,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Lersivirine (UK-453061) is a next-generation non-nucleoside inhibitor of reverse transcriptase under development for the treatment of HIV-1 infection. | |||||
| Targets | reverse transcriptase | |||||

Lersivirine Dilution Calculator

Lersivirine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2222 mL | 16.1108 mL | 32.2217 mL | 64.4434 mL | 80.5542 mL |
| 5 mM | 0.6444 mL | 3.2222 mL | 6.4443 mL | 12.8887 mL | 16.1108 mL |
| 10 mM | 0.3222 mL | 1.6111 mL | 3.2222 mL | 6.4443 mL | 8.0554 mL |
| 50 mM | 0.0644 mL | 0.3222 mL | 0.6444 mL | 1.2889 mL | 1.6111 mL |
| 100 mM | 0.0322 mL | 0.1611 mL | 0.3222 mL | 0.6444 mL | 0.8055 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Lersivirine (UK-453061) is a next-generation non-nucleoside reverse transcriptase inhibitor (NNRTI)for human immunodeficincy virus (HIV) infection with IC50 value of 119 nM [1].
HIV is a retro virus that causes HIV infection and acquired immunodeficiency syndrome (AIDS). It may infect vital cells of human immune system such as helper T cells and dendritic cells. HIV transcriptase plays an important role in the infection process. HIV carries a reverse transcriptase which can transcript single-stranded virus RNA into double-stranded DNA. When the virus anchor to the target cell surface, the reverse transcriptase will be injected into host cell, there it may complete the transcription. And the transcribed DNA is able to integrate into host genome to complete infection and viral replication.
Lersivirine (UK-453061) is a NNRTI with a unique resistance profile that exhibits potent antiretroviral activity against wild-type HIV and clinically relevant NNRTI-resistant strains. When lersivirine was tested with a panel of isolated wild-type and drug-resistant HIV reverse transcriptase, it exhibited excellent inhibitory activity, which confirmed the high potency of it as the next-generation anti-HIV NNRTI. The compound also has good aqueous solubility and formulation characteristics which enable further in vivo evaluation [2].
Mated Crl:CD1 mice were administered 0, 150, 350, and 500 mg/kg lersivirine once daily by oral gavage on gestation days 6 to 17, followed by cesarean section on gestation day 18. The first 2 days of dosing for the high-dose group were done at 250 mg/kg to allow induction of hepatic metabolizing enzymes, after which the dose was increased to 500 mg/kg/day. Exposure of lersivirine did not cause any increases in external, visceral, or skeletal malformation, which demonstrated lersivirine is not teratogenic in mice [3].
References:
[1] Mowbray C E et al. , Pyrazole NNRTIs 4: selection of UK-453,061 (lersivirine) as a development candidate. Bioorg Med Chem Lett. 2009, 19(20):5857-60.
[2] Davis J et al. , The effect of lersivirine, a next-generation NNRTI, on the pharmacokinetics of midazolam and oral contraceptives in healthy subjects. Eur J Clin Pharmacol. 2012, 68(11):1567-1572.
[3] Cappon G D et al. , Developmental toxicity study of lersivirine in mice. Birth Defects Res B Dev Reprod Toxicol. 2012, 95(3):225-30.
- Boc-Tyr(tBu)-OH
Catalog No.:BCC3462
CAS No.:47375-34-8
- SCH 563705
Catalog No.:BCC1933
CAS No.:473728-58-4
- SCH 527123
Catalog No.:BCC1932
CAS No.:473727-83-2
- AMG 487
Catalog No.:BCC5140
CAS No.:473719-41-4
- Boc-Trp(For)-OH
Catalog No.:BCC3456
CAS No.:47355-10-2
- Carpinontriol B
Catalog No.:BCN8113
CAS No.:473451-73-9
- Spegatrine
Catalog No.:BCN4068
CAS No.:47326-53-4
- Betulin
Catalog No.:BCN5528
CAS No.:473-98-3
- Tolbutamide Sodium
Catalog No.:BCC5632
CAS No.:473-41-6
- beta-Eudesmol
Catalog No.:BCN6294
CAS No.:473-15-4
- alpha-Cyperone
Catalog No.:BCN1193
CAS No.:473-08-5
- SB 366791
Catalog No.:BCC7128
CAS No.:472981-92-3
- Forskolin G
Catalog No.:BCN5527
CAS No.:473981-11-2
- Brazilin
Catalog No.:BCN5529
CAS No.:474-07-7
- Chenodeoxycholic acid
Catalog No.:BCN2620
CAS No.:474-25-9
- Citrostadienol
Catalog No.:BCN7357
CAS No.:474-40-8
- Reserpin N-oxide
Catalog No.:BCN3493
CAS No.:474-48-6
- Daucosterol
Catalog No.:BCN5531
CAS No.:474-58-8
- Campestanol
Catalog No.:BCN3890
CAS No.:474-60-2
- Campesterol
Catalog No.:BCN3181
CAS No.:474-62-4
- Brassicasterol
Catalog No.:BCN2613
CAS No.:474-67-9
- CAL-130 Racemate
Catalog No.:BCC1442
CAS No.:474012-90-3
- N-Methylsarpagine methosalt
Catalog No.:BCN5530
CAS No.:47418-70-2
- Monomethyl auristatin E
Catalog No.:BCC1775
CAS No.:474645-27-7
Effects of lersivirine on canine and rodent thyroid function.[Pubmed:24135463]
Toxicol Pathol. 2014 Jul;42(5):897-912.
Lersivirine is a nonnucleoside reverse transcriptase inhibitor (NNRTI) being developed for the treatment of HIV-1 infection. Like other NNRTIs, Lersivirine is a potent enzyme inducer in rodents capable of inducing a number of hepatic enzymes including those involved in its own metabolism. Preclinically Lersivirine has been associated with hepatocellular hypertrophy and thyroid gland follicular cell hypertrophy in rats, mice, and dogs. In rodents, we show that development of thyroid hypertrophy is related to the classic mechanism, namely increased thyroxine (T4) clearance secondary to induction of uridine-diphosphoglucuronosyltransferase (UDPGT) in the liver and a resulting increase in thyroid-stimulating hormone. Similarly, Lersivirine-exposed dogs exhibit a significant increase in hepatic UDPGT enzyme activity along with increased T4 clearance although clear effects on serum thyroid hormone levels were less apparent. These effects on thyroid hormonal clearance in the dog suggest that thyroid gland hypertrophy in this species is due to the same mechanism shown to occur in rodents although, as expected, dogs better adapt to these effects and therefore maintain relatively normal thyroid hormonal balance. It is also notable that the minimal thyroid follicular hypertrophy that occurs in dogs does not progress as is seen in rodents. As is the case with rodents, these adaptive changes in the dog are not considered indicative of a human health risk.
Efficacy and safety of lersivirine versus etravirine for the treatment of HIV-1 infection in patients with prior non-nucleoside reverse transcriptase inhibitor (NNRTI) use and evidence of NNRTI resistance: a randomized phase 2B trial.[Pubmed:25350959]
HIV Clin Trials. 2014 Sep-Oct;15(5):209-17.
OBJECTIVE: To assess the efficacy and safety of Lersivirine versus etravirine in patients with HIV-1 and prior non-nucleoside reverse transcriptase inhibitor (NNRTI) use and evidence of NNRTI resistance. METHODS: In this 96-week, phase 2b study, 97 patients were randomized and treated with Lersivirine 750 mg qd (n = 31), Lersivirine 1,000 mg qd (n = 32), and etravirine 200 mg bid (n = 34), plus one optimized nucleoside reverse transcriptase inhibitor and darunavir/ritonavir 600/100 mg bid. The primary endpoint was the percentage of patients with HIV-1 RNA <50 copies/mL at week 24. RESULTS: At week 24, HIV-1 RNA <50 copies/mL was achieved by fewer patients receiving Lersivirine 750 mg (48.4%) and 1,000 mg (43.8%) qd compared with etravirine 200 mg qd (67.7%) (intention to treat [ITT], missing/switch/discontinuation equals failure [MSDF]). At week 48, HIV-1 RNA <50 copies/mL and <400 copies/mL were also achieved by fewer patients receiving Lersivirine 750 mg (41.9% and 41.9%, respectively) and 1,000 mg (31.3% and 34.4%, respectively) qd compared with etravirine 200 mg bid (61.8% and 70.6%, respectively) (ITT, MSDF). Least squares means (SE) change from baseline in log transformed HIV-1 RNA at week 48 was -1.42 (0.27) and -0.95 (0.28) copies/mL for Lersivirine 750 mg and 1,000 mg qd, respectively, versus -2.02 (0.26) copies/mL for etravirine 200 mg bid (ITT). Lersivirine and etravirine were generally safe and well-tolerated. CONCLUSIONS: Lersivirine 750 mg and 1,000 mg qd was associated with lower rates of viral suppression at week 24 and week 48 versus etravirine in patients with prior NNRTI use and evidence of NNRTI resistance. No new safety signals were detected.
Lersivirine - a new drug for HIV infection therapy.[Pubmed:24128277]
Expert Opin Investig Drugs. 2013 Dec;22(12):1687-94.
INTRODUCTION: Lersivirine (UK-453,061) is a novel second-generation non-nucleoside reverse transcriptase inhibitor (NNRTI). It binds reverse transcriptase in a distinct way leading to a unique resistance profile. The development of Lersivirine was recently stopped in Phase IIb clinical development. AREAS COVERED: This article describes the background of Lersivirine, its pharmacodynamic and pharmacokinetic profile and its clinical efficacy in HIV-infected patients. Moreover, the authors review its resistance profile in addition to its possible interactions with coadministered drugs and safety and tolerability. The authors' evaluation is based on the articles retrieved from a Medline in addition to abstracts from major HIV conferences and workshops addressing Lersivirine. EXPERT OPINION: The authors believe that Lersivirine has therapeutic potential for HIV-infected individuals with viral strains resistant against first-line NNRTIs. However, no large, well-powered studies have been conducted so far, which assess noninferiority against established antiretroviral agents. In February 2013, the developing company behind Lersivirine halted further development as it was decided that it would not provide an improvement over existing therapies; perhaps this is an opportunity missed.


