NAS-181Selective r5-HT1B antagonist. Active in vivo CAS# 1217474-40-2 |
- MK-5172 hydrate
Catalog No.:BCC1763
CAS No.:1350462-55-3
- MK-5172 sodium salt
Catalog No.:BCC1765
CAS No.:1425038-27-2
- Telaprevir (VX-950)
Catalog No.:BCC2107
CAS No.:402957-28-2
- Asunaprevir (BMS-650032)
Catalog No.:BCC1374
CAS No.:630420-16-5
- Danoprevir (RG7227)
Catalog No.:BCC2106
CAS No.:850876-88-9
- Vaniprevir
Catalog No.:BCC2030
CAS No.:923590-37-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1217474-40-2 | SDF | Download SDF |
| PubChem ID | 45073445 | Appearance | Powder |
| Formula | C21H34N2O10S2 | M.Wt | 538.63 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in water and to 100 mM in DMSO | ||
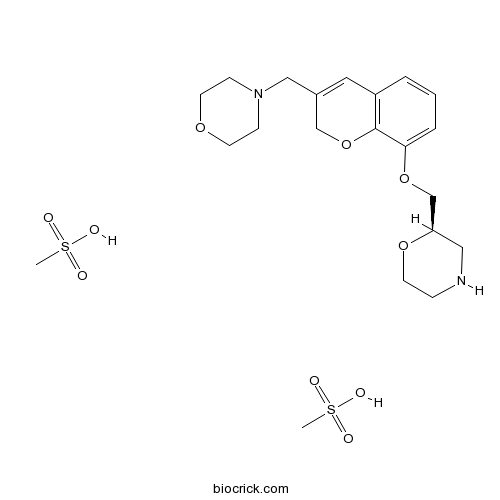
| Chemical Name | methanesulfonic acid;(2R)-2-[[3-(morpholin-4-ylmethyl)-2H-chromen-8-yl]oxymethyl]morpholine | ||
| SMILES | CS(=O)(=O)O.CS(=O)(=O)O.C1COC(CN1)COC2=CC=CC3=C2OCC(=C3)CN4CCOCC4 | ||
| Standard InChIKey | WMRMIRRDPBKNMY-ZEECNFPPSA-N | ||
| Standard InChI | InChI=1S/C19H26N2O4.2CH4O3S/c1-2-16-10-15(12-21-5-8-22-9-6-21)13-25-19(16)18(3-1)24-14-17-11-20-4-7-23-17;2*1-5(2,3)4/h1-3,10,17,20H,4-9,11-14H2;2*1H3,(H,2,3,4)/t17-;;/m1../s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent, selective antagonist at the rat 5-HT1B receptor (Ki = 47 nM). Increases synthesis and metabolism of 5-HT in the brain following systemic administration and improves passive avoidance retention performance in vivo. Increases subthalamic nucleus-triggered complex EPSCs and burst firing in SNr GABA neurons |

NAS-181 Dilution Calculator

NAS-181 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8566 mL | 9.2828 mL | 18.5656 mL | 37.1312 mL | 46.4141 mL |
| 5 mM | 0.3713 mL | 1.8566 mL | 3.7131 mL | 7.4262 mL | 9.2828 mL |
| 10 mM | 0.1857 mL | 0.9283 mL | 1.8566 mL | 3.7131 mL | 4.6414 mL |
| 50 mM | 0.0371 mL | 0.1857 mL | 0.3713 mL | 0.7426 mL | 0.9283 mL |
| 100 mM | 0.0186 mL | 0.0928 mL | 0.1857 mL | 0.3713 mL | 0.4641 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- (+)-UH 232 maleate
Catalog No.:BCC6790
CAS No.:1217473-50-1
- Isochandalone
Catalog No.:BCN4767
CAS No.:121747-90-8
- Isoderrone
Catalog No.:BCN3698
CAS No.:121747-89-5
- Ro 26-4550 trifluoroacetate
Catalog No.:BCC5813
CAS No.:1217448-66-2
- VD2-D3
Catalog No.:BCC2034
CAS No.:1217448-46-8
- A 350619 hydrochloride
Catalog No.:BCC5939
CAS No.:1217201-17-6
- CP 31398 dihydrochloride
Catalog No.:BCC2406
CAS No.:1217195-61-3
- 6-O-p-Methoxycinnamoylcatalpol
Catalog No.:BCN6109
CAS No.:121710-02-9
- GYKI 47261 dihydrochloride
Catalog No.:BCC7566
CAS No.:1217049-32-5
- BU 239 hydrochloride
Catalog No.:BCC5668
CAS No.:1217041-98-9
- PTC209 HBr
Catalog No.:BCC5640
CAS No.:1217022-63-3
- Moluccanin diacetate
Catalog No.:BCN6108
CAS No.:121700-27-4
- threo-1-C-Syringylglycerol
Catalog No.:BCN6110
CAS No.:121748-11-6
- BYL-719
Catalog No.:BCC3707
CAS No.:1217486-61-7
- SB 205607 dihydrobromide
Catalog No.:BCC5687
CAS No.:1217628-73-3
- SB 258719 hydrochloride
Catalog No.:BCC5937
CAS No.:1217674-10-6
- RS 16566 dihydrochloride
Catalog No.:BCC6890
CAS No.:1217788-97-0
- 5-Iodo-A-85380 dihydrochloride
Catalog No.:BCC7099
CAS No.:1217837-17-6
- Ac-Trp-OH
Catalog No.:BCC3109
CAS No.:1218-34-4
- Xylometazoline HCl
Catalog No.:BCC4879
CAS No.:1218-35-5
- Pidotimod
Catalog No.:BCC4823
CAS No.:121808-62-6
- CAY10505
Catalog No.:BCC4990
CAS No.:1218777-13-9
- LDE225 Diphosphate
Catalog No.:BCC1693
CAS No.:1218778-77-8
- GKT137831
Catalog No.:BCC5460
CAS No.:1218942-37-0
Facilitation and inhibition of male rat ejaculatory behaviour by the respective 5-HT1A and 5-HT1B receptor agonists 8-OH-DPAT and anpirtoline, as evidenced by use of the corresponding new and selective receptor antagonists NAD-299 and NAS-181.[Pubmed:9886765]
Br J Pharmacol. 1998 Dec;125(8):1733-43.
1. Ejaculatory problems and anorgasmia are well-known side-effects of the SSRI antidepressants, and a pharmacologically induced increase in serotonergic neurotransmission inhibits ejaculatory behaviour in the rat. In the present study the role of 5-HT1A and 5-HT1B receptors in the mediation of male rat ejaculatory behaviour was examined by use of selective agonists and antagonists acting at these 5-HT receptor subtypes. 2. The 5-HT1A receptor agonist 8-OH-DPAT (0.25-4.00 micromol kg(-1) s.c.) produced an expected facilitation of the male rat ejaculatory behaviour, and this effect was fully antagonized by pretreatment with the new selective 5-HT1A receptor antagonist (R)-3-N,N-dicyclobutylamino-8-fluoro-3,4-dihydro-2H-1-benzopyran-5 -carboxamide hydrogen (2R,3R) tartrate monohydrate (NAD-299) (1.0 micromol kg(-1) s.c.). NAD-299 by itself (0.75-3.00 micromol kg(-1) s.c.) did not affect the male rat ejaculatory behaviour. 3. The 5-HT1B receptor agonist anpirtoline (0.25-4.00 micromol kg(-1) s.c.) produced a dose-dependent inhibition of the male rat ejaculatory behaviour, and this effect was fully antagonized by pretreatment with the 5-HT1B receptor antagonist isamoltane (16 micromol kg(-1) s.c.) as well as by the new and selective antagonist (R)-(+)-2-(3-morpholinomethyl-2H-chromene-8-yl)oxymethylmorphol ino methansulphonate (NAS-181) (16 micromol kg(-1) s.c.). Isamoltane (1.0-16.0 micromol kg(-1) s.c.) and NAD-181 (1.0-16.0 micromol kg(-1) s.c.) had no, or weakly facilitatory effects on the male rat ejaculatory behaviour. The non-selective 5-HT1 receptor antagonist (-)-pindolol (8 micromol kg(-1) s.c.), did not antagonize the inhibition produced by anpirtoline. 4. The present results demonstrate opposite effects, facilitation and inhibition, of male rat ejaculatory behaviour by stimulation of 5-HT1A and 5-HT1B receptors, respectively, suggesting that the SSRI-induced inhibition of male ejaculatory dysfunction is due to 5-HT1B receptor stimulation.
An evaluation of the effect of NAS-181, a new selective 5-HT(1B) receptor antagonist, on extracellular 5-HT levels in rat frontal cortex.[Pubmed:12595948]
Naunyn Schmiedebergs Arch Pharmacol. 2003 Feb;367(2):89-94.
In the mammalian brain 5-HT(1B) receptors are present as autoreceptors regulating the release of serotonin (5-HT) by inhibitory feedback. The antagonistic properties of NAS-181 ((R)-(+)-2-[[[3-(Morpholinomethyl)-2H-chromen-8-yl]oxy]methyl] morpholine methane sulfonate), a new selective antagonist for the rodent 5-HT(1B) receptor, were determined by using an agonist-induced decrease of extracellular 5-HT. The 5-HT(1B) receptor agonist CP93129 (0.030.3 microM) applied by reversed microdialysis, dose-dependently reduced 5-HT levels in rat frontal cortex. The suppressant effect of CP93129 (0.1 microM) was smaller in the presence of fluvoxamine (3-10 microM), a 5-HT reuptake inhibitor. The effects of NAS-181 on CP93129 were compared with GR127935, a mixed 5-HT (1B/1D) receptor antagonist, and SB224289, a 5-HT(1B) receptor antagonist. Both in the presence and absence of fluvoxamine, the suppressant effect of CP93129 on extracellular 5-HT was attenuated by NAS-181 (1 microM) and GR127935 (10 microM), but not by SB224289 (1 microM). In the absence of fluvoxamine, GR127935, SB224289 and NAS-181 all reduced 5-HT levels, suggesting partial agonistic properties of these compounds. In conclusion, the results show that NAS-181 is a potent 5-HT(1B) receptor antagonist.
Enhanced 5-HT metabolism and synthesis rate by the new selective r5-HT1B receptor antagonist, NAS-181 in the rat brain.[Pubmed:10728876]
Neuropharmacology. 2000 Feb 14;39(4):553-60.
NAS-181 ((R)-(+)-2-(3-morpholinomethyl-2H-chromen-8-yl) oxymethyl-morpholine methanesulfonate) is a novel rat 5-hydroxytryptamine1B, (r5-HT1B) receptor antagonist with high selectivity. The in vivo effects of NAS-181 on 5-HT metabolism and synthesis in the rat brain were examined. 5-HT metabolism, measured as the ratio 5-hydroxyindoleacetic acid (5-HIAA)/5-HT, was dose-dependently increased in all four brain regions analysed (hypothalamus, hippocampus, frontal cortex and striatum) at doses 0.1 to 20 mg/kg s.c. NAS-181. The enhancement of 5-HT metabolism at the dose 20 mg/kg s.c. was maximal one hour after the injection and was still significant eight hours but not 24 hours after the injection. 5-HT synthesis rate measured as the accumulation of 5-hydroxytryptophan (5-HTP) after inhibition of the aromatic amino acid decarboxylase activity was also elevated by NAS-181 at doses 0.3 to 20 mg/kg s.c. NAS-181 competitively antagonised the decrease in 5-HT metabolism evoked by the r5-HT1B receptor agonist, anpirtoline, in hypothalamus, hippocampus and frontal cortex. Anpirtoline had no effect on 5-HT metabolism in striatum. However, anpirtoline antagonised the enhancement of 5-HT metabolism induced by NAS-181 in striatum. Combined treatment of rats with NAS-181 and the 5-HT1A receptor antagonist, WAY-100635, increased 5'-HT metabolism considerably more than when the compounds were given alone.
Effects of the 5-HT1B receptor antagonist NAS-181 on extracellular levels of acetylcholine, glutamate and GABA in the frontal cortex and ventral hippocampus of awake rats: a microdialysis study.[Pubmed:17234388]
Eur Neuropsychopharmacol. 2007 Sep;17(9):580-6.
The purpose of this study was to investigate the effects of the 5-HT(1B) receptor antagonist NAS-181 ((R)-(+)-2-(3-morpholinomethyl-2H-chromen-8-yl) oxymethyl-morpholine methanesulfonate) on cholinergic, glutamatergic and GABA-ergic neurotransmission in the rat brain in vivo. Extracellular levels of acetylcholine, glutamate and GABA were monitored by microdialysis in the frontal cortex (FC) and ventral hippocampus (VHipp) in separate groups of freely moving rats. NAS-181 (1, 5 or 10 mg/kg, s.c.) caused a dose-dependent increase in ACh levels, reaching the maximal values of 500% (FC) and 230% (VHipp) of controls at 80 min post-injection. On the contrary, NAS-181 injected at doses of 10 or 20 mg/kg s.c. had no effect on basal extracellular levels of Glu and GABA in these areas. The present data suggest that ACh neurotransmission in the FC and VHipp, the brain structures strongly implicated in cognitive function, is under tonic inhibitory control of 5-HT(1B) heteroreceptors localized at the cholinergic terminals in these areas.
Presynaptic serotonergic gating of the subthalamonigral glutamatergic projection.[Pubmed:23486958]
J Neurosci. 2013 Mar 13;33(11):4875-85.
The GABAergic projection neurons in the substantia nigra pars reticulata (SNr) are key basal ganglia output neurons. The activity of these neurons is critically influenced by the glutamatergic projection from the subthalamic nucleus (STN). The SNr also receives an intense serotonin (5-HT) innervation, raising the possibility that 5-HT may regulate the STN-->SNr glutamatergic transmission and the consequent STN-triggered spike firing in SNr neurons. Here we show that 5-HT reduced STN stimulation-evoked long-lasting polysynaptic complex EPSCs in SNr GABA neurons. This inhibitory 5-HT effect was mimicked by the 5-HT1B receptor agonist CP93129 and blocked by the 5-HT1B antagonist NAS-181. 5-HT1A receptor ligands were ineffective. Additionally, 5-HT and CP93129 reduced the frequency but not the amplitude of miniature EPSCs, suggesting a reduced vesicular release. 5-HT and CP93129 also decreased the amplitude but increased the paired pulse ratio of the monosynaptic EPSCs in SNr GABA neurons, indicating a presynaptic 5-HT1B receptor-mediated inhibition of glutamate release. Furthermore, 5-HT and CP93129 inhibited STN-triggered burst firing in SNr GABA neurons, and CP93129's inhibitory effect was strongest when puffed to STN-->SNr axon terminals in SNr, indicating a primary role of the 5-HT1B receptors in these axon terminals. Finally, the 5-HT1B receptor antagonist NAS-181 increased the STN-triggered complex EPSCs and burst firing in SNr GABA neurons, demonstrating the effects of endogenous 5-HT. These results suggest that nigral 5-HT, via presynaptic 5-HT1B receptor activation, gates the excitatory STN-->SNr projection, reduces burst firing in SNr GABA neurons, and thus may play a critical role in movement control.
Blockade of 5-HT 1B receptors facilitates contextual aversive learning in mice by disinhibition of cholinergic and glutamatergic neurotransmission.[Pubmed:18394658]
Neuropharmacology. 2008 Jun;54(7):1041-50.
Serotonergic (5-HT) neurotransmission plays a role in learning and memory processes, but the physiological role of various receptor subtypes is not well characterised. Among these, 5-HT(1B) receptors are located as autoreceptors on 5-HT axons and heteroreceptors on non-serotonergic terminals. This study examined the role of the 5-HT(1B) receptor in one-trial aversive contextual learning using the passive avoidance (PA) task in NMRI mice. Subcutaneous administration of the 5-HT(1B) receptor agonist anpirtoline (0.1-1.0mg/kg) before PA training impaired retention performance 24h later. Combined administration of anpirtoline with the selective 5-HT(1B) receptor antagonist NAS-181 (0.1-1.0mg/kg) fully blocked the impairments. Administration of NAS-181 alone dose-dependently improved PA retention performance. This facilitatory effect was blocked by subthreshold doses of both the muscarinic antagonist scopolamine (0.03 mg/kg) and the NMDA receptor antagonist MK-801 (0.03 mg/kg). NAS-181 also fully blocked the PA impairments induced by an amnesic dose of scopolamine (0.1mg/kg), when administered prior to, but not after, scopolamine. In addition, NAS-181 attenuated PA impairments induced by MK-801 (0.3mg/kg). These findings indicate that 5-HT(1B) receptors are activated at basal levels of 5-HT transmission. The facilitatory effect of NAS-181 involved alleviation of an inhibitory 5-HT tone mediated via 5-HT(1B) receptors on cholinergic and glutamatergic transmission. This disinhibition is expected to occur in neuronal circuits involved in contextual learning including the hippocampus and interconnected cortico-limbic regions. Blockade of brain 5-HT(1B) heteroreceptors may represent a novel therapeutic strategy for restoration of deficient cholinergic and glutamatergic neurotransmission contributing to memory disorders.
(R)-(+)-2-[[[3-(Morpholinomethyl)-2H-chromen-8-yl]oxy]methyl] morpholine methanesulfonate: a new selective rat 5-hydroxytryptamine1B receptor antagonist.[Pubmed:9599242]
J Med Chem. 1998 May 21;41(11):1934-42.
In the search for new 5-hydroxytryptamine (5-HT) receptor antagonists it was found that the compound (R)-(+)-2-[[[3-(morpholinomethyl)-2H-chromen-8-yl]oxy] methyl] morpholine methanesulfonate, (R)-25, is a selective rat 5-hydroxytryptamine1B (r5-HT1B) receptor antagonist. The binding profile showed a 13-fold preference for r5-HT1B (Ki = 47 +/- 5 nM; n = 3) vs bovine 5-HT1B (Ki = 630 nM; n = 1) receptors. The compound had very low affinity for other monoaminergic receptors examined. The r5-HT1B receptor antagonism was demonstrated by the potentiation of the K+-stimulated release of [3H]-5-HT from superfused rat brain slices in vitro, an effect that was antagonized by addition of 5-HT to the superfusion fluid. (R)-25 at 20 mg/kg sc enhanced the 5-HT turnover in four rat brain regions (hypothalamus, hippocampus, striatum, and frontal cortex) with about 40% measured as the 5-HTP accumulation after decarboxylase inhibition with 3-hydroxybenzylhydrazine. At 3 mg/kg sc (R)-25 produced a significant increase in the number of wet dog shakes in rats, a 5-HT2A/5-HT2C response that was abolished by depletion of 5-HT after pretreatment with the tryptophan hydroxylase inhibitor p-chlorophenylalanine. These observations show that (R)-25, by inhibiting terminal r5-HT1B autoreceptors, increases the 5-HT turnover and the synaptic concentration of 5-HT.


