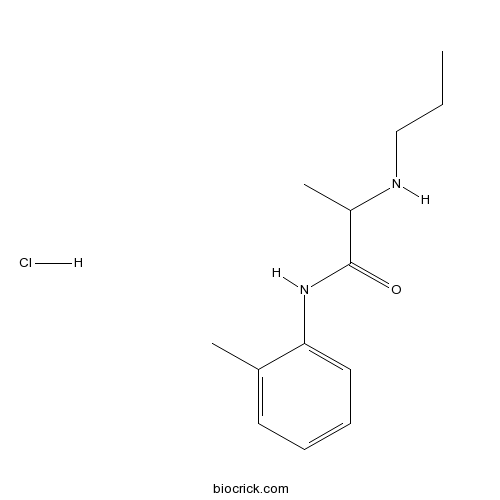
Prilocaine hydrochlorideCAS# 1786-81-8 |
- Istradefylline (KW-6002)
Catalog No.:BCC3798
CAS No.:155270-99-8
- ANR 94
Catalog No.:BCC7815
CAS No.:634924-89-3
- Tozadenant
Catalog No.:BCC2011
CAS No.:870070-55-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1786-81-8 | SDF | Download SDF |
| PubChem ID | 92163 | Appearance | Powder |
| Formula | C13H21ClN2O | M.Wt | 256.77 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | >10.4mg/mL in DMSO | ||
| Chemical Name | N-(2-methylphenyl)-2-(propylamino)propanamide;hydrochloride | ||
| SMILES | CCCNC(C)C(=O)NC1=CC=CC=C1C.Cl | ||
| Standard InChIKey | BJPJNTKRKALCPP-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C13H20N2O.ClH/c1-4-9-14-11(3)13(16)15-12-8-6-5-7-10(12)2;/h5-8,11,14H,4,9H2,1-3H3,(H,15,16);1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Prilocaine hydrochloride is a local anesthetic of the amino amide type.
Target: Others
Prilocaine is a local anesthetic of the amino amide type first prepared by Claes Tegner and Nils L?fgren. In its injectable form (trade name Citanest), it is often used in dentistry. It is also often combined with lidocaine as a preparation for dermal anesthesia, for treatment of conditions like paresthesia. As it has low cardiac toxicity, it is commonly used for intravenous regional anaesthesia (IVRA). In some patients, a metabolite of prilocaine may cause the unusual side effect of methemoglobinemia, which may be treated with methylene blue. Maximum dosage for dental use: 8.0 mg/kg (2.7 mg/lb), with a maximum dose of 500 mg.
Eutectic Mixture of Local Anesthetics (EMLA) containing 5% lidocaine and prilocaine in a cream was found to give effective topical analgesia in normal and diseased skin, making it useful for superficial surgery and various other clinical procedures. To be effective, an adequate amount must be applied under occlusion and at the right time before the intervention. References: | |||||

Prilocaine hydrochloride Dilution Calculator

Prilocaine hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.8945 mL | 19.4727 mL | 38.9454 mL | 77.8907 mL | 97.3634 mL |
| 5 mM | 0.7789 mL | 3.8945 mL | 7.7891 mL | 15.5781 mL | 19.4727 mL |
| 10 mM | 0.3895 mL | 1.9473 mL | 3.8945 mL | 7.7891 mL | 9.7363 mL |
| 50 mM | 0.0779 mL | 0.3895 mL | 0.7789 mL | 1.5578 mL | 1.9473 mL |
| 100 mM | 0.0389 mL | 0.1947 mL | 0.3895 mL | 0.7789 mL | 0.9736 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Prilocaine hydrochloride is a local anesthetic of the amino amide type.
- Hoechst 33342 analog
Catalog No.:BCC1630
CAS No.:178481-68-0
- Vitexin -4''-O-glucoside
Catalog No.:BCN3054
CAS No.:178468-00-3
- 6-epi-Albrassitriol
Catalog No.:BCN7342
CAS No.:178456-58-1
- Agrostophyllidin
Catalog No.:BCN3598
CAS No.:178439-50-4
- AR-R 17779 hydrochloride
Catalog No.:BCC7827
CAS No.:178419-42-6
- Tos-Arg-OMe.HCl
Catalog No.:BCC2874
CAS No.:1784-03-8
- 12-Hydroxy-6-epi-albrassitriol
Catalog No.:BCN7460
CAS No.:178330-78-4
- H-D-Asp-OH
Catalog No.:BCC2894
CAS No.:1783-96-6
- Nociceptin (1-7)
Catalog No.:BCC5738
CAS No.:178249-42-8
- Orphanin FQ (1-11)
Catalog No.:BCC6085
CAS No.:178249-41-7
- Calystegine B3
Catalog No.:BCN1880
CAS No.:178231-95-3
- Tetrahymanone
Catalog No.:BCN6932
CAS No.:17822-06-9
- ZD 2079
Catalog No.:BCC5878
CAS No.:178600-17-4
- Oleoside
Catalog No.:BCN1134
CAS No.:178600-68-5
- U-104
Catalog No.:BCC2312
CAS No.:178606-66-1
- CFM-2
Catalog No.:BCC6931
CAS No.:178616-26-7
- rel-(8R,8'R)-dimethyl-(7S,7'R)-bis(3,4-methylenedioxyphenyl)tetrahydro-furan
Catalog No.:BCN1521
CAS No.:178740-32-4
- Agrocybenine
Catalog No.:BCN4755
CAS No.:178764-92-6
- Delta3,2-Hydroxylbakuchiol
Catalog No.:BCN3707
CAS No.:178765-49-6
- 3-Hydroxybakuchiol
Catalog No.:BCN3610
CAS No.:178765-54-3
- 12-Hydroxyisobakuchiol
Catalog No.:BCN3609
CAS No.:178765-55-4
- SNC 162
Catalog No.:BCC7103
CAS No.:178803-51-5
- Vitexolide D
Catalog No.:BCN6739
CAS No.:1788090-69-6
- Myricadiol
Catalog No.:BCN1135
CAS No.:17884-88-7
Prilocaine hydrochloride protects zebrafish from lethal effects of ionizing radiation: role of hematopoietic cell expansion.[Pubmed:25843444]
Tokai J Exp Clin Med. 2015 Mar 20;40(1):8-15.
Drug repositioning is an approach of significant translatability, and the present study was undertaken to screen a collection of FDA approved small-molecule clinical compounds for identification of novel radioprotective agents. Screening of JHCCL (Johns Hopkins Clinical Compound Library), a collection of 1,400 FDA approved small molecules, lead to identification of Prilocaine hydrochloride, a local anesthetic used widely during dental procedures, as a potential radioprotector. Prilocaine, at a concentration of 20 microM, protected zebrafish from radiation induced (20 Gy) pericardial edema (PE), microphthalmia and rendered 60 % survival advantage over radiation exposed controls. While 40 % survival advantage over radiation exposed controls was achieved with 10 microM prilocaine. Prilocaine, in a dose-dependent manner, scavenged, radiation-induced hydroxyl radicals and maximally (43 %) at the highest concentration (1 mM) tried in this study. However, prilocaine exerted a mild superoxide anion scavenging potential (around 5 %) at all the concentrations used within this study. Prilocaine, at 20 microM concentration, significantly increased erythropoiesis, a marker for HSC function, in caudal hematopoietic tissue (CHT) in wild type and anemic zebrafish embryos (1.48 and 0.85 folds respectively) when compared to untreated (1) and phenylhydrazine (PHZ) (0.41 fold) treated control groups respectively. These results suggest that prilocaine is a radioprotective agent and free radical scavenging and HSC expanding potential seems to be contributing towards its radioprotective action.
Verification of protector effect of the norepinephrine and felypressin upon the cardiovascular system under action of the lidocaine hydrochloride and prilocaine hydrochloride in anesthetized rats.[Pubmed:12220948]
Pharmacol Res. 2002 Aug;46(2):107-11.
Vasoconstrictor substances, as norepinephrine and epinephrine, were mixtured to local anesthetics to decrease their toxic effects and to prolong the depth of the anesthesia. However, these catecholamines produce systolic and diastolic hypertension. The effects of felypressin, a synthetic vasoconstrictor, upon arterial blood pressure and heart are lesser than those of norepinephrine or epinephrine, but due to its effects like oxytocin these catecholamines are yet the most used vasoconstrictors in association with lidocaine or another anesthetic salt. These vasoconstrictors are contraindicated for some physician, mainly for cardiac patients. But, are the catecholamines or is the salt the most dangerous components of the local anesthetic? The effects of the salt and catecholamines are opposite, but which of these exercises their effects first when inside blood vessel? Singi et al. [Pharmacol. Res. 44 (2001)] demonstrated that the first effect is always of the salt and that norepinephrine promotes protector effects upon guinea-pig isolated heart against lidocaine action. But, is this true for in vivo animals? The present study was performed with the aiming to answer this question and to verify if felypressin can induce the same effect of the norepinephrine. Fourteen Rattus norvegicus albinus, weighing 350g on average, were used. After being anesthetized with sodic thiopental, they were tracheostomizeds and one jugular and one carotid were cannuled for application of substances and to record the blood arterial pressure, respectively. The ECG was gotten through electrodes located in the front and back paws of the animals. The animals were separated in two groups, each one with seven rats. The lidocaine hydrochloride 2% in the doses of 600 microg and 3% in the doses of 900 microg acted on the cardiovascular system reducing the arterial pressure and modifying the electrocardiogram, while the Prilocaine hydrochloride, in the same doses, also reduced the arterial pressure, but did not modify the electrocardiogram. When norepinephrine was associated to lidocaine 3% hydrochloride, it was possible to observe that this salt always exercised its effect first and a protective effect against the fall of pressure produced for the lidocaine. The same protective effect did not occur when felypressin was associated with Prilocaine hydrochloride 3%.


