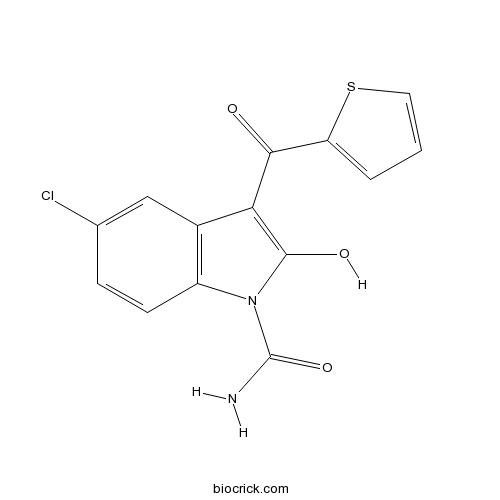
TenidapNSAID, cyclooxygenase (COX-1) inhibitor. Also opener of KIR2.3 CAS# 120210-48-2 |
- AZD8055
Catalog No.:BCC3629
CAS No.:1009298-09-2
- WYE-354
Catalog No.:BCC1059
CAS No.:1062169-56-5
- GDC-0349
Catalog No.:BCC1094
CAS No.:1207360-89-1
- Everolimus (RAD001)
Catalog No.:BCC3594
CAS No.:159351-69-6
- Rapamycin (Sirolimus)
Catalog No.:BCC3592
CAS No.:53123-88-9
- KU-0063794
Catalog No.:BCC2484
CAS No.:938440-64-3
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 120210-48-2 | SDF | Download SDF |
| PubChem ID | 60712 | Appearance | Powder |
| Formula | C14H9N2O3SCl | M.Wt | 320.75 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | CP 66248 | ||
| Solubility | Soluble to 100 mM in DMSO | ||
| Chemical Name | (Z)-5-Chloro-2,3-dihydro-3-(hydroxy | ||
| SMILES | NC(=O)n1c(O)c(C(=O)c2sccc2)c3cc(Cl)ccc13 | ||
| Standard InChIKey | IZSFDUMVCVVWKW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C14H9ClN2O3S/c15-7-3-4-9-8(6-7)11(13(19)17(9)14(16)20)12(18)10-2-1-5-21-10/h1-6,19H,(H2,16,20) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | NSAID that preferentially inhibits COX-1 (IC50 values are < 0.03, 1.2 and > 30 μM for COX-1, COX-2 and 5-lipoxygenase respectively). Inhibits formation of pro-inflammatory arachidonic acid metabolites in isolated human peripheral polymorphonuclear leukocytes. Opener of inward rectifying hKir2.3 channel (EC50 = 402 nM). |

Tenidap Dilution Calculator

Tenidap Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1177 mL | 15.5885 mL | 31.1769 mL | 62.3539 mL | 77.9423 mL |
| 5 mM | 0.6235 mL | 3.1177 mL | 6.2354 mL | 12.4708 mL | 15.5885 mL |
| 10 mM | 0.3118 mL | 1.5588 mL | 3.1177 mL | 6.2354 mL | 7.7942 mL |
| 50 mM | 0.0624 mL | 0.3118 mL | 0.6235 mL | 1.2471 mL | 1.5588 mL |
| 100 mM | 0.0312 mL | 0.1559 mL | 0.3118 mL | 0.6235 mL | 0.7794 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- NMS-P715
Catalog No.:BCC6373
CAS No.:1202055-34-2
- Clopidogrel Related Compound C
Catalog No.:BCN2689
CAS No.:120202-71-3
- Clopidogrel
Catalog No.:BCC2497
CAS No.:120202-66-6
- 3,4-Dihydroxycinnamamide
Catalog No.:BCN6090
CAS No.:1202-41-1
- Cynoglossamine
Catalog No.:BCN1970
CAS No.:120193-39-7
- TCS 2210
Catalog No.:BCC7798
CAS No.:1201916-31-5
- MLN9708
Catalog No.:BCC2091
CAS No.:1201902-80-8
- Vinflunine Tartrate
Catalog No.:BCC4602
CAS No.:1201898-17-0
- Crotaleschenine
Catalog No.:BCN2077
CAS No.:120154-95-2
- IPI-145 (INK1197)
Catalog No.:BCC1104
CAS No.:1201438-56-3
- Flavanthrin
Catalog No.:BCN3687
CAS No.:120090-80-4
- Fmoc-D-Arg(Mtr)-OH
Catalog No.:BCC3078
CAS No.:120075-24-3
- 1beta-Hydroxyeuscaphic acid
Catalog No.:BCN3517
CAS No.:120211-98-5
- CGS 21680
Catalog No.:BCC1475
CAS No.:120225-54-9
- AVL-292
Catalog No.:BCC1385
CAS No.:1202757-89-8
- CNX-774
Catalog No.:BCC4394
CAS No.:1202759-32-7
- Salvinolone
Catalog No.:BCN3215
CAS No.:120278-22-0
- 4-Hydroxysapriparaquinone
Catalog No.:BCN4806
CAS No.:120278-25-3
- Dorzolamide
Catalog No.:BCC4287
CAS No.:120279-96-1
- CX-6258
Catalog No.:BCC1504
CAS No.:1202916-90-2
- Citroside A
Catalog No.:BCN7294
CAS No.:120330-44-1
- DASA-58
Catalog No.:BCC6522
CAS No.:1203494-49-8
- Cyclotraxin B
Catalog No.:BCC6357
CAS No.:1203586-72-4
- AS 1949490
Catalog No.:BCC7762
CAS No.:1203680-76-5
Opposing effects of tenidap on the volume-regulated anion channel and K(ATP) channel activity in rat pancreatic beta-cells.[Pubmed:20035738]
Eur J Pharmacol. 2010 Mar 10;629(1-3):159-63.
Tenidap (5-chloro-2-hydroxy-3-(thiophene-2-carbonyl)indole-1-carboxamide) is a non-steroidal anti-inflammatory and anti-rheumatic drug with several cellular actions including inhibition of anion transport processes. Since other anion transport inhibitors have been shown to inhibit activity of the volume-regulated anion channel (VRAC), the present study investigated the effects of Tenidap on activity of this channel in pancreatic beta-cells. Membrane potential, VRAC currents and input conductance were recorded from single rat beta-cells in primary culture using perforated patch, conventional whole-cell and cell-attached configurations of the patch-clamp technique. Relative cell volume was measured using a video-imaging method. Tenidap (0.1mM) was found to rapidly hyperpolarise the beta-cell membrane potential and terminate glucose-induced electrical activity. This effect was associated with a pronounced outward current shift at a holding potential of -65mV. Tenidap was found to inhibit activity of the volume-regulated anion channel with IC(50) values of 31 and 43microM for outward and inward currents respectively. Tenidap also markedly increased beta-cell input conductance, representing an activation of the K(ATP) conductance. beta-cell regulatory volume decrease following hypotonically-induced cell swelling was sensitive to inhibition by 50microM Tenidap. Tenidap is a potent inhibitor of the volume-regulated anion channel and K(ATP) channel activator in rat pancreatic beta-cells. These actions could at least in part explain the recently reported inhibitory actions of the drug on electrical and secretory activity in the beta-cell, and could also underlie other pharmacological actions of the drug.
Tenidap is neuroprotective in a pilocarpine rat model of temporal lobe epilepsy.[Pubmed:23673107]
Chin Med J (Engl). 2013;126(10):1900-5.
BACKGROUND: Tenidap is a liposoluble non-steroidal anti-inflammatory drug that is easily distributed in the central nervous system and also inhibits the production and activity of cyclooxygenase-2 (COX-2) and cytokines in vitro. This study aimed to evaluate the neuroprotective effect of Tenidap in a pilocarpine rat model of temporal lobe epilepsy (TLE). METHODS: Tenidap was administered daily at 10 mg/kg for 10 days following pilocarpine-induced status epilepticus (SE) in male Wistar rats after which prolonged generalized seizures resulted in TLE. After Tenidap treatment, spontaneous recurrent seizures (SRSs) were recorded by video monitoring (for 7 hours per day for 14 days). The frequency and severity of the SRSs were observed. Histological and immunocytochemical analyses were used to evaluate the neuroprotective effect of Tenidap and detect COX-2 expression, which may be associated with neuronal death. RESULTS: There were 46.88 +/- 10.70 survival neurons in Tenidap-SE group, while there were 27.60 +/- 5.18 survival neurons in saline-SE group at -2.4 mm field in the CA3 area. There were 37.75 +/- 8.78 survival neurons in Tenidap-SE group, while there were 33.40 +/- 8.14 survival neurons in saline-SE group at -2.4 mm field in the CA1 area. Tenidap treatment significantly reduced neuronal damage in the CA3 area (P < 0.05) and slightly reduced damage in the CA1 area. Tenidap markedly inhibited COX-2 expression in the hippocampus, especially in the CA3 area. CONCLUSION: Tenidap conferred neuroprotection to the CA3 area in a pilocarpine-induced rat model of TLE by inhibiting COX-2 expression.
Tenidap, an agonist of the inwardly rectifying K+ channel Kir2.3, delays the onset of cortical epileptiform activity in a model of chronic temporal lobe epilepsy.[Pubmed:23561319]
Neurol Res. 2013 Jul;35(6):561-7.
OBJECTIVE: Inwardly rectifying K(+) (Kir) channel, Kir2.3, has been found down-regulated in the hippocampus of chronic temporal lobe epileptic (cTLE) patients which may underline the mechanism of epilepsy. Tenidap is an agonist of Kir2.3. However, the effect of Tenidap on cTLE remains obscure. The aim of the present study was to observe the relationship between Kir2.3 and the pathogenesis of cTLE, and to explore the potential effect of Kir agonists as anti-epileptic drugs. METHODS: The pilocarpine temporal lobe epilepsy (TLE) rat model was established and status epilepticus (SE) was induced in rats. The rats were divided into four groups based on time points (0 hours, 6 hours, 72 hours, and two weeks) after SE termination. The dynamic changes in Kir2.3 mRNA and protein expression in the hippocampus were detected at each time point by reverse transcription polymerase chain reaction (RT-PCR) and western blotting to determine an appropriate time for further intervention and observation. Then, Tenidap was administered and its effect on the expression of Kir2.3 mRNA/protein and the electroencephalogram (EEG) wave was documented at predetermined time points. RESULTS: The pattern of Kir2.3 mRNA and protein expression was bimodal, as it increased immediately after SE and declined at two weeks, when compared with the control group. Down-regulation of the Kir channels may lead to an impaired clearance of K(+) ions from the extracellular space and result in a stronger neuronal depolarisation. Two weeks after SE may be a reversal point. Spontaneous recurrent seizures (SRS) appeared at the two-week point, indicating that chronic epilepsy had occurred. Tenidap up-regulated expressions of both Kir2.3 channel mRNA and protein, which may help to maintain the resting membrane potential (RMP) and hyperpolarisation of the cells. CONCLUSIONS: Our results suggest that the down-regulation of the Kir2.3 channel expression might contribute to the pathogenesis of TLE, which may be ameliorated by the administration of Tenidap.
A comparison of the disease-modifying and cytokine-regulating activities of tenidap, piroxicam and cyclosporin-A using the adjuvant-induced model of arthritis in rats.[Pubmed:17657619]
Inflammopharmacology. 1998;6(3):193-202.
This study compared the antiarthritic activity of Tenidap, piroxicam and cyclosporin-A (CsA) using the model of adjuvant-induced arthritis in rats. The aim of the study was to correlate any disease-modifying effects of Tenidap with its in-vivo regulation of cytokines.Both Tenidap and piroxicam reduced arthritic disease when administered orally from the time the first signs of arthritis are expressed. Disease suppression correlated with a significant reduction in interleukin-6 production and a slight reduction in interleukin-1 and tumour necrosis factor production. When coadministered with the adjuvant, Tenidap and CsA prevented disease in 50% and 100% of animals, respectively, whereas piroxicam had no effect. This disease prevention induced by Tenidap and CsA coincided with reduced interferon-gamma and interleukin-2 production by lymph node cells one day following initiation of adjuvant disease. This inhibition of T-cell cytokines might be consistent with Tenidap acting as a disease-modifying drug.
Evaluation of the antiinflammatory activity of a dual cyclooxygenase-2 selective/5-lipoxygenase inhibitor, RWJ 63556, in a canine model of inflammation.[Pubmed:9262379]
J Pharmacol Exp Ther. 1997 Aug;282(2):1094-101.
Sterile perforated polyethylene spheres (wiffle golf balls) were implanted s.c. in beagle dogs. A local inflammatory reaction was elicited within the spheres by injecting carrageenan. Changes in leukocyte count, prostaglandin E2, thromboxane B2 and leukotriene B4 levels were monitored in fluid samples collected over a 24-hr period. Blood samples were also collected at various time points and analyzed for prostaglandin E2 and leukotriene B4 production after ex vivo calcium ionophore treatment. Effects of standard antiinflammatory agents (aspirin, indomethacin, dexamethasone, Tenidap and zileuton) and newer cyclooxygenase-2 (COX-2) selective agents (nimesulide, nabumetone and SC-58125) were determined after oral administration. Ex vivo inhibition of cyclooxygenase product synthesis (prostaglandin E2, thromboxane B2) in whole blood was used as an indicator of activity for the constitutive COX-1 isoform, although inhibition of the synthesis of these mediators in the chamber exudate during an inflammatory process is believed to represent COX-2 inhibition. Treatment effects on leukotriene B4 production were also determined both ex vivo in whole blood and in the fluid. All of the compounds tested, except aspirin, inhibited leukocyte infiltration into the fluid exudate. Inhibitors that exert their effects on both isozymes of cyclooxygenase attenuate production of cyclooxygenase metabolites in both the inflammatory exudate and in peripheral blood ex vivo, although COX-2 selective inhibitors only demonstrated activity in the exudate. A 5-lipoxygenase inhibitor (zileuton), a corticosteroid (dexamethasone) and a dual COX-2 selective/5-lipoxygenase inhibitor (RWJ 63556) had similar profiles in that they all inhibited cell infiltration and eicosanoid production in the fluid and also attenuated leukotriene B4 production in both the fluid and blood.
CP-66,248, a new anti-inflammatory agent, is a potent inhibitor of leukotriene B4 and prostanoid synthesis in human polymorphonuclear leucocytes in vitro.[Pubmed:2856170]
Eicosanoids. 1988;1(1):35-9.
The effects of (Z)-5-chloro-2,3-dihydro-3-(hydroxy-2-thienylmethylene)-2-oxo-1H- indole-1-carboxamide (CP-66,248), a new anti-inflammatory agent, were tested on the synthesis of the pro-inflammatory arachidonic acid metabolites, LTB4 and PGE2, in isolated human peripheral polymorphonuclear leucocytes. At clinically achievable (i.e. plasma) drug concentration, CP-66,248 reduced A 23187-stimulated LTB4 (IC50 18 +/- 1 microM) and PGE2 (IC50 32 +/- 8 nM) synthesis. The corresponding IC50 values for arachidonic acid-induced LTB4 and PGE2 production were 13 +/- 4 microM and 65 +/- 15 nM, respectively. The inhibitory action of CP-66,248 towards 5-lipoxygenase was comparable with that of timegadine and exceeded that of caffeic acid, and its action against the cyclo-oxygenase pathway was similar to that of other NSAIDs tested. The dual inhibition of cyclo-oxygenase and lipoxygenase pathways of arachidonic acid metabolism is likely to be involved in the anti-inflammatory, antipyretic and analgetic action of CP-66,248 detected in a variety of experimental models.


