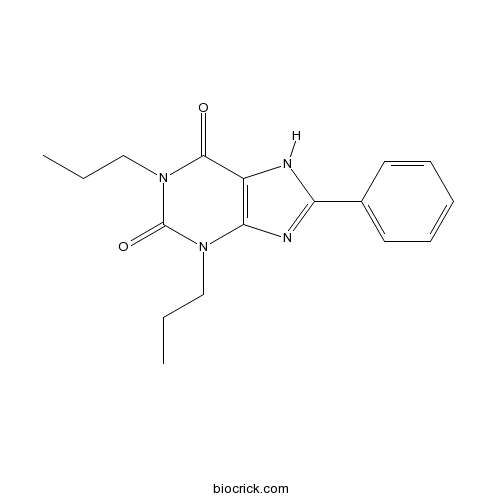
1,3-Dipropyl-8-phenylxanthineA1 selective antagonist CAS# 85872-53-3 |
- CYT387 sulfate salt
Catalog No.:BCC1506
CAS No.:1056636-06-6
- Baricitinib phosphate
Catalog No.:BCC1401
CAS No.:1187595-84-1
- JAK2 Inhibitor V, Z3
Catalog No.:BCC1667
CAS No.:195371-52-9
- Bardoxolone methyl
Catalog No.:BCC1400
CAS No.:218600-53-4
- Ruxolitinib (INCB018424)
Catalog No.:BCC1276
CAS No.:941678-49-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 85872-53-3 | SDF | Download SDF |
| PubChem ID | 128784 | Appearance | Powder |
| Formula | C17H20N4O2 | M.Wt | 312.37 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 5 mM in ethanol with gentle warming and to 10 mM in DMSO with gentle warming | ||
| Chemical Name | 8-phenyl-1,3-dipropyl-7H-purine-2,6-dione | ||
| SMILES | CCCN1C2=C(C(=O)N(C1=O)CCC)NC(=N2)C3=CC=CC=C3 | ||
| Standard InChIKey | CLIGSMOZKDCDRZ-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H20N4O2/c1-3-10-20-15-13(16(22)21(11-4-2)17(20)23)18-14(19-15)12-8-6-5-7-9-12/h5-9H,3-4,10-11H2,1-2H3,(H,18,19) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective A1 adenosine antagonist. |

1,3-Dipropyl-8-phenylxanthine Dilution Calculator

1,3-Dipropyl-8-phenylxanthine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2013 mL | 16.0067 mL | 32.0133 mL | 64.0266 mL | 80.0333 mL |
| 5 mM | 0.6403 mL | 3.2013 mL | 6.4027 mL | 12.8053 mL | 16.0067 mL |
| 10 mM | 0.3201 mL | 1.6007 mL | 3.2013 mL | 6.4027 mL | 8.0033 mL |
| 50 mM | 0.064 mL | 0.3201 mL | 0.6403 mL | 1.2805 mL | 1.6007 mL |
| 100 mM | 0.032 mL | 0.1601 mL | 0.3201 mL | 0.6403 mL | 0.8003 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Marsdenoside F
Catalog No.:BCN4564
CAS No.:858360-61-9
- Vialinin A
Catalog No.:BCC2367
CAS No.:858134-23-3
- 3-O-Benzyl estrone
Catalog No.:BCC8638
CAS No.:858-98-0
- (-)-Quinpirole hydrochloride
Catalog No.:BCC6917
CAS No.:85798-08-9
- 11,12-Di-O-acetyltenacigenin B
Catalog No.:BCN4565
CAS No.:857897-01-9
- Motesanib Diphosphate (AMG-706)
Catalog No.:BCC2477
CAS No.:857876-30-3
- Polygalaxanthone XI
Catalog No.:BCN7366
CAS No.:857859-82-6
- Alstolenine
Catalog No.:BCN4808
CAS No.:85769-33-1
- Longistylumphylline A
Catalog No.:BCN4408
CAS No.:857672-34-5
- PSN632408
Catalog No.:BCC5408
CAS No.:857652-30-3
- AT7867
Catalog No.:BCC2536
CAS No.:857531-00-1
- K-252c
Catalog No.:BCC3706
CAS No.:85753-43-1
- prim-O-Glucosylangelicain
Catalog No.:BCN4409
CAS No.:85889-15-2
- Lincomycin hydrochloride
Catalog No.:BCC9011
CAS No.:859-18-7
- 6-Iodonordihydrocapsaicin
Catalog No.:BCC5860
CAS No.:859171-97-4
- 4-O-Methylhonokiol
Catalog No.:BCN8474
CAS No.:68592-15-4
- Acetyl meldrum's acid
Catalog No.:BCC8805
CAS No.:85920-63-4
- AGN 205728
Catalog No.:BCC5418
CAS No.:859498-05-8
- GR 125487 sulfamate
Catalog No.:BCC7142
CAS No.:859502-43-5
- FIT
Catalog No.:BCC7082
CAS No.:85951-63-9
- BMS-690514
Catalog No.:BCC1430
CAS No.:859853-30-8
- Anemosapogenin
Catalog No.:BCN2454
CAS No.:85999-40-2
- Carbazole
Catalog No.:BCN6903
CAS No.:86-74-8
- Benzoyleneurea
Catalog No.:BCC8865
CAS No.:86-96-4
[3H]xanthine amine congener of 1,3-dipropyl-8-phenylxanthine: an antagonist radioligand for adenosine receptors.[Pubmed:3012550]
Proc Natl Acad Sci U S A. 1986 Jun;83(11):4089-93.
An amine-functionalized derivative of 1,3-Dipropyl-8-phenylxanthine has been prepared in tritiated form as a xanthine amine congener ([3H]XAC) for use as an antagonist radioligand for adenosine receptors. [3H]XAC has higher receptor affinity, higher specific activity, lower nonspecific membrane binding, and more favorable hydrophilicity than 1,3-diethyl-8-[3H]phenylxanthine, the xanthine commonly used for adenosine receptor binding. In rat cerebral cortical membranes, [3H]XAC exhibits saturable, specific binding with a Kd of 1.23 nM and a Bmax of 580 fmol/mg of protein at 37 degrees C. N6-(R-Phenylisopropyl)adenosine is a more potent inhibitor of [3H]XAC binding than is 5'-N-ethylcarboxamidoadenosine, indicating that binding is to an A1-adenosine receptor. In the absence of GTP, the inhibition curves for adenosine agonists versus [3H]XAC binding are biphasic, indicating that [3H]XAC is binding to low- and high-affinity agonist states of the A1 receptor. In the presence of GTP, adenosine analogs exhibit monophasic, low-affinity inhibition of binding of [3H]XAC. Inhibition of [3H]XAC binding by theophylline or by various 8-phenylxanthines is monophasic, and the potencies are commensurate with the potencies of these xanthines as adenosine receptor antagonists. The receptor sites in calf brain membranes exhibit a higher affinity (Kd = 0.17 nM) for [3H]XAC, whereas sites in guinea pig exhibit a slightly lower affinity (Kd = 3.0 nM). Densities of [3H]XAC binding sites are similar in brain membranes from all species.
[Selectivity of the action of 1,3-dipropyl-8-phenylxanthine on the purine receptors of the intestines].[Pubmed:3191974]
Farmakol Toksikol. 1988 Jul-Aug;51(4):60-2.
In experiments on the isolated ileum of guinea pigs and rats it was shown that 1,3-Dipropyl-8-phenylxanthine (DPPX) is a highly potent adenosine antagonist blocking A1-adenosine receptors of the myenteric plexus (pA2 = 7.29 +/- 0.06) and allosterically modulating affinity of A2-receptors of the intestinal smooth muscles to adenosine (pA2 = 6.64 +/- 0.05). DPPX exerted no influence on purinergic (PR2), adrenergic and opiate receptors of the nervous and muscular cells of the intestine. Exceeding the activity of theophylline by 339-87 times, DPPX failed to exert the selective effect on A1- and A2-subtypes of adenosine receptors.
Analogues of 1,3-dipropyl-8-phenylxanthine: enhancement of selectivity at A1-adenosine receptors by aryl substituents.[Pubmed:3016270]
J Med Chem. 1986 Aug;29(8):1520-4.
The effect of a variety of aryl substituents on the potency and selectivity of 19 analogues of 1,3-Dipropyl-8-phenylxanthine as antagonists at A1- and A2-adenosine receptors in brain tissue was determined. The 4-sulfamoylphenyl and 4-carbamoylphenyl analogues are potent and somewhat selective for the A1 receptor. None of the dihydroxyphenyl analogues are remarkably potent, but all are selective for the A1 receptor. 1,3-Dipropyl-8-(2-hydroxy-4-methoxyphenyl)xanthine is the most selective A1 antagonist of the analogues with a A1/A2 potency ratio of about 90.
A [3H]amine congener of 1,3-dipropyl-8-phenylxanthine. A new radioligand for A2 adenosine receptors of human platelets.[Pubmed:3009222]
FEBS Lett. 1986 Apr 21;199(2):269-74.
A xanthine amine congener (XAC), an amine-functionalized derivative of 1,3-Dipropyl-8-phenylxanthine, is an antagonist ligand for A2 adenosine receptors of human platelets. XAC inhibited 5'-N-ethylcarboxamidoadenosine (NECA)-induced stimulation of adenylate cyclase activity with a KB of 24 nM. [3H]XAC exhibits saturable, specific binding with a Kd of 12 nM and a Bmax of 1.1 pmol/mg protein at 37 degrees C. [3H]XAC binding in platelets is the first example of labeling of A2 adenosine receptors in which the potencies of adenosine agonists and antagonists in inhibiting binding are commensurate with their potencies at these receptors in functional studies. Furthermore, [3H]XAC is the first antagonist radioligand with high affinity at A2 adenosine receptors.
1,3-Dialkyl-8-(p-sulfophenyl)xanthines: potent water-soluble antagonists for A1- and A2-adenosine receptors.[Pubmed:2984420]
J Med Chem. 1985 Apr;28(4):487-92.
A series of 8-(substituted phenyl) derivatives of theophylline and other 1,3-dialkylxanthines were evaluated for potency and selectivity as antagonists at A1- and A2-adenosine receptors in brain tissue. Theophylline has a similar potency (Ki = 14 microM) at both A1 and A2 receptors. 8-Phenyltheophylline is 25-35-fold more potent as an adenosine receptor antagonist than theophylline, while 8-phenylcaffeine is only 2-3-fold more potent than caffeine. A p-hydroxyaryl substituent enhances the potency of 8-phenyltheophylline as an adenosine antagonist. p-Carboxy- and p-sulfoaryl substituents reduce potency of 8-phenyltheophylline, yielding water-soluble adenosine antagonists, which are some 2-5-fold more potent than theophylline at adenosine receptors. None of the 8-(substituted phenyl)theophyllines are particularly selective as antagonists toward A1- and A2-adenosine receptors. 1,3-Dipropyl-8-phenylxanthine represents a potent and somewhat selective A1-receptor antagonist about 23-fold more potent at A1 receptors than at A2 receptors. A p-hydroxyaryl substituent further enhances potency of the 1,3-Dipropyl-8-phenylxanthine at both A1 and A2 receptors. The 8-(2-amino-4-chlorophenyl)-1,3-dipropylxanthine is a very potent and selective antagonist for A1 receptors, being nearly 400-fold more potent at A1 than at A2 receptors. The water-soluble 8-(p-sulfophenyl)- and 8-(p-carboxyphenyl)-1,3-propylxanthines no longer exhibit marked selectivity. Both compounds are much more potent as adenosine antagonists than theophylline. The striking selectivity of 1-isoamyl-3-isobutylxanthine as an A1 antagonist is retained in the 8-phenyl derivative but is virtually lost in the 8-p-sulfophenyl derivative.


