B-Raf inhibitorA B-Raf inhibitor CAS# 1315330-11-0 |
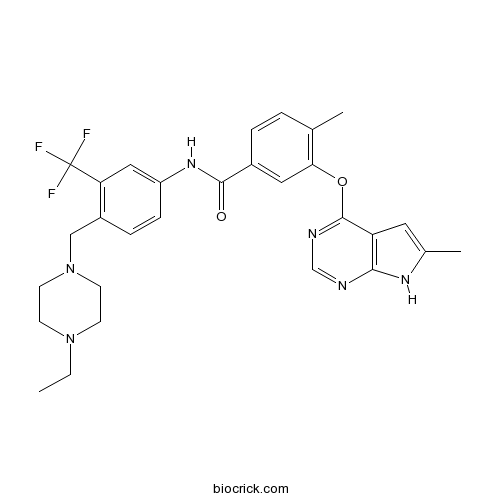
- CEP-32496
Catalog No.:BCC1079
CAS No.:1188910-76-0
- Sorafenib
Catalog No.:BCN2174
CAS No.:284461-73-0
- Vemurafenib (PLX4032, RG7204)
Catalog No.:BCC1269
CAS No.:918504-65-1
- BRAF inhibitor
Catalog No.:BCC1436
CAS No.:918505-61-0
- PLX-4720
Catalog No.:BCC1280
CAS No.:918505-84-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1315330-11-0 | SDF | Download SDF |
| PubChem ID | 71254032 | Appearance | Powder |
| Formula | C29H31F3N6O2 | M.Wt | 552.59 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 50 mg/mL (90.48 mM; Need ultrasonic) | ||
| Chemical Name | N-[4-[(4-ethylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl]-4-methyl-3-[(6-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-yl)oxy]benzamide | ||
| SMILES | CCN1CCN(CC1)CC2=C(C=C(C=C2)NC(=O)C3=CC(=C(C=C3)C)OC4=NC=NC5=C4C=C(N5)C)C(F)(F)F | ||
| Standard InChIKey | RWNAOXLCVXJMGM-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C29H31F3N6O2/c1-4-37-9-11-38(12-10-37)16-21-7-8-22(15-24(21)29(30,31)32)36-27(39)20-6-5-18(2)25(14-20)40-28-23-13-19(3)35-26(23)33-17-34-28/h5-8,13-15,17H,4,9-12,16H2,1-3H3,(H,36,39)(H,33,34,35) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | A B-Raf inhibitor, pyrazine and pyrrolo[2,3-b]pyridine derivatives, useful in the treatment of cancer and proliferative diseases. References: | |||||

B-Raf inhibitor Dilution Calculator

B-Raf inhibitor Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8097 mL | 9.0483 mL | 18.0966 mL | 36.1932 mL | 45.2415 mL |
| 5 mM | 0.3619 mL | 1.8097 mL | 3.6193 mL | 7.2386 mL | 9.0483 mL |
| 10 mM | 0.181 mL | 0.9048 mL | 1.8097 mL | 3.6193 mL | 4.5241 mL |
| 50 mM | 0.0362 mL | 0.181 mL | 0.3619 mL | 0.7239 mL | 0.9048 mL |
| 100 mM | 0.0181 mL | 0.0905 mL | 0.181 mL | 0.3619 mL | 0.4524 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
A B-Raf inhibitor, pyrazine and pyrrolo[2,3-b]pyridine derivatives, useful in the treatment of cancer and proliferative diseases.
- HG6-64-1
Catalog No.:BCC5459
CAS No.:1315329-43-1
- TMP269
Catalog No.:BCC3993
CAS No.:1314890-29-3
- Amyloid Beta-Peptide (1-40) (human)
Catalog No.:BCC1045
CAS No.:131438-79-4
- Cercosporamide
Catalog No.:BCC2438
CAS No.:131436-22-1
- UNC669
Catalog No.:BCC3997
CAS No.:1314241-44-5
- TC-N 22A
Catalog No.:BCC6150
CAS No.:1314140-00-5
- Hemopressin (human, mouse)
Catalog No.:BCC6065
CAS No.:1314035-51-2
- CBZ-Osu
Catalog No.:BCC2798
CAS No.:13139-17-8
- Boc-Ile-OH.1/2H2O
Catalog No.:BCC3406
CAS No.:13139-16-7
- Boc-Leu-OH.H2O
Catalog No.:BCC3408
CAS No.:13139-15-6
- Boc-Trp-OH
Catalog No.:BCC3455
CAS No.:13139-14-5
- Teijin compound 1
Catalog No.:BCC6057
CAS No.:1313730-14-1
- NG25
Catalog No.:BCC1799
CAS No.:1315355-93-1
- pep2-EVKI
Catalog No.:BCC5786
CAS No.:1315378-67-6
- pep2-AVKI
Catalog No.:BCC5787
CAS No.:1315378-69-8
- TCS 184
Catalog No.:BCC5899
CAS No.:1315378-71-2
- Scrambled 10Panx
Catalog No.:BCC1246
CAS No.:1315378-72-3
- PDZ1 Domain inhibitor peptide
Catalog No.:BCC5883
CAS No.:1315378-73-4
- Bax inhibitor peptide, negative control
Catalog No.:BCC2395
CAS No.:1315378-74-5
- MNI caged kainic acid
Catalog No.:BCC7297
CAS No.:1315378-75-6
- pep2-SVKE
Catalog No.:BCC5785
CAS No.:1315378-76-7
- Rac1 Inhibitor F56, control peptide
Catalog No.:BCC5887
CAS No.:1315378-77-8
- NPEC-caged-(1S,3R)-ACPD
Catalog No.:BCC7653
CAS No.:1315379-60-2
- VU 591 hydrochloride
Catalog No.:BCC6126
CAS No.:1315380-70-1
New insights into renal toxicity of the B-RAF inhibitor, vemurafenib, in patients with metastatic melanoma.[Pubmed:27371224]
Cancer Chemother Pharmacol. 2016 Aug;78(2):419-26.
PURPOSE: Vemurafenib (VMF) is a B-Raf inhibitor used in the treatment of B-RAF-V600-mutant metastatic melanomas. Reports of acute kidney injury (AKI) in patients treated with VMF are scarce. METHODS: To investigate the incidence and severity of AKI, we conducted a retrospective, observational, monocentric study in the Lyon Sud Hospital University, France, which included 74 patients with metastatic B-RAF-mutated melanomas treated with VMF, between June 2011 and August 2014. According to the Kidney Disease Improving Global Outcomes Guidelines, AKI is defined as an increase in serum creatinine concentration exceeding the baseline concentration by 1.5 fold. Serum creatinine was thus determined before treatment, on a monthly basis during treatment, and 3 months after treatment discontinuation. Patients were divided into two main groups: AKI-positive (AKI+) and AKI-negative (AKI-) and further subdivided into three groups according to AKI severity (stage 1, 2 or 3). To visualize the tissue damage caused by VMF, kidney biopsies were performed for two stage 1 AKI+ patients. RESULTS: Of the 74 patients, 30 (40.5 %) were AKI-, and of the 44 AKI+ patients (59.5 %), 29 (66 %) were diagnosed within the first three months of treatment. There were significantly more men in the AKI+ group: n = 33 (75 %) versus n = 12 (40 %) women, p = 0.004 with an odds ratio for developing AKI of 4.6 (95 % CI 1.48-14.23). Most AKI + cases were considered as stage 1 (n = 40; 91 %) and the remaining four (9 %) as stage 2 AKI. Kidney biopsies revealed interstitial fibrosis and acute focal tubular damage. However, renal failure was reversible in 80 % of patients within 3 months of VMF discontinuation. CONCLUSIONS: We observed frequent, reversible, moderately severe AKI with some histological evidence of tubular and interstitial damage in VMF-treated patients, suggesting that renal function should be carefully monitored in male patients, especially during the first 3 months.
Determination of a novel B-Raf(V600E) and EGFR dual inhibitor in rat plasma by HPLC-MS/MS and its application in a pharmacokinetic study.[Pubmed:27423011]
J Pharm Biomed Anal. 2016 Sep 10;129:142-147.
The EGFR and B-Raf(V600E) dual inhibition is a promising strategy in treatment of colorectal cancer patients with B-Raf(V600E) mutation. Previously, compound 3 was designed and synthesized as a novel B-Raf(V600E) and EGFR dual inhibitor with highly potency in both kinase and cell based assay. Herein, a sensitive and rapid HPLC-MS/MS quantitative method was developed and validated for the further pharmacokinetic evaluation of compound 3 in rats.


