Ethyl rosmarinateCAS# 174591-47-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 174591-47-0 | SDF | Download SDF |
| PubChem ID | 44437692.0 | Appearance | Powder |
| Formula | C20H20O8 | M.Wt | 388.37 |
| Type of Compound | Phenylpropanoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
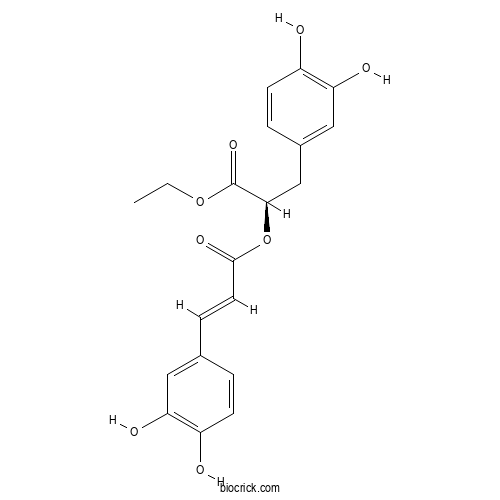
| Chemical Name | ethyl (2R)-3-(3,4-dihydroxyphenyl)-2-[(E)-3-(3,4-dihydroxyphenyl)prop-2-enoyl]oxypropanoate | ||
| SMILES | CCOC(=O)C(CC1=CC(=C(C=C1)O)O)OC(=O)C=CC2=CC(=C(C=C2)O)O | ||
| Standard InChIKey | ROJRNQOAUDCMES-KRZKXXONSA-N | ||
| Standard InChI | InChI=1S/C20H20O8/c1-2-27-20(26)18(11-13-4-7-15(22)17(24)10-13)28-19(25)8-5-12-3-6-14(21)16(23)9-12/h3-10,18,21-24H,2,11H2,1H3/b8-5+/t18-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Ethyl rosmarinate Dilution Calculator

Ethyl rosmarinate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5749 mL | 12.8743 mL | 25.7486 mL | 51.4973 mL | 64.3716 mL |
| 5 mM | 0.515 mL | 2.5749 mL | 5.1497 mL | 10.2995 mL | 12.8743 mL |
| 10 mM | 0.2575 mL | 1.2874 mL | 2.5749 mL | 5.1497 mL | 6.4372 mL |
| 50 mM | 0.0515 mL | 0.2575 mL | 0.515 mL | 1.0299 mL | 1.2874 mL |
| 100 mM | 0.0257 mL | 0.1287 mL | 0.2575 mL | 0.515 mL | 0.6437 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3,5,7-Trimethoxyflavone
Catalog No.:BCX1280
CAS No.:26964-29-4
- Micromarin F
Catalog No.:BCX1279
CAS No.:73292-93-0
- Palvanil
Catalog No.:BCX1278
CAS No.:69693-13-6
- N-Acetylcytisine
Catalog No.:BCX1277
CAS No.:6018-52-6
- Hispidol
Catalog No.:BCX1276
CAS No.:5786-54-9
- 2',4,4',6'-Tetramethoxychalcone
Catalog No.:BCX1275
CAS No.:94103-36-3
- 2'-O-Methylphloretin
Catalog No.:BCX1274
CAS No.:111316-17-7
- Phenoxodiol
Catalog No.:BCX1273
CAS No.:81267-65-4
- 6-Hydroxyluteolin
Catalog No.:BCX1272
CAS No.:18003-33-3
- Sinomenine N-oxide
Catalog No.:BCX1271
CAS No.:1000026-77-6
- Protoanemonin
Catalog No.:BCX1270
CAS No.:108-28-1
- Demethyldaphnoretin-7-O-glucoside
Catalog No.:BCX1269
CAS No.:438578-91-7
- 1-(2,6-Dimethoxyphenyl)-3-(4-hydroxyphenyl)-2-propen-1-one
Catalog No.:BCX1282
CAS No.:85679-87-4
- 5-Hydroxy-3,7-dimethoxyflavone
Catalog No.:BCX1283
CAS No.:70786-48-0
- Morusignin L
Catalog No.:BCX1284
CAS No.:149733-95-9
- Napelline
Catalog No.:BCX1285
CAS No.:5008-52-6
- Isobellidifolin
Catalog No.:BCX1286
CAS No.:552-00-1
- Dihydrocatalpol
Catalog No.:BCX1287
CAS No.:6736-86-3
- Ajugose
Catalog No.:BCX1288
CAS No.:512-72-1
- Limocitrin 3-O-β-D-glucopyranoside
Catalog No.:BCX1289
CAS No.:38836-51-0
- Wistin
Catalog No.:BCX1290
CAS No.:19046-26-5
- Physcion-8-O-β-gentiobioside
Catalog No.:BCX1291
CAS No.:84268-38-2
- Chaparrinone
Catalog No.:BCX1292
CAS No.:22611-34-3
- Ajugasterone C 2-acetate
Catalog No.:BCX1293
CAS No.:154510-93-7
Effects of rosmarinic acid esters on the oxidation kinetic of organogel and emulsion gel.[Pubmed:38586224]
Food Chem X. 2024 Mar 31;22:101343.
Rosmarinic acid was esterified with ethanol, butanol, and hexanol to produce Ethyl rosmarinate, butyl rosmarinate, and hexyl rosmarinate, respectively. The antioxidant capacities of the rosmarinic acid esters were evaluated in linseed oil, organogel, and emulsion gel during the initiation and propagation phases of peroxidation. Organogel control sample showed higher induction period and propagation period than those of linseed oil and emulsion gel control samples. Among linseed oil and organogel samples containing antioxidants, samples containing rosmarinic acid exhibited the highest antioxidant activity during the initiation phase, while rosemary extract containing butyl rosmarinate showed the highest antioxidant activity in the propagation phase. In emulsion gel, rosemary extract containing butyl rosmarinate showed higher antioxidant activity than those of rosemary extract containing Ethyl rosmarinate or hexyl rosmarinate in the initiation and propagation phases. In addition, the investigated antioxidants showed lower efficiency in organogel and emulsion gel samples than those in linseed oil samples.


