LY2795050KOR antagonist CAS# 1346133-08-1 |
- Alvimopan monohydrate
Catalog No.:BCC1349
CAS No.:1383577-62-5
- Alvimopan
Catalog No.:BCC1347
CAS No.:156053-89-3
- Alvimopan dihydrate
Catalog No.:BCC1348
CAS No.:170098-38-1
- ADL5859 HCl
Catalog No.:BCC1265
CAS No.:850173-95-4
- Cebranopadol
Catalog No.:BCC1467
CAS No.:863513-91-1
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1346133-08-1 | SDF | Download SDF |
| PubChem ID | 56851583 | Appearance | Powder |
| Formula | C23H22ClN3O2 | M.Wt | 407.89 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 50 mg/mL (122.58 mM; Need ultrasonic) | ||
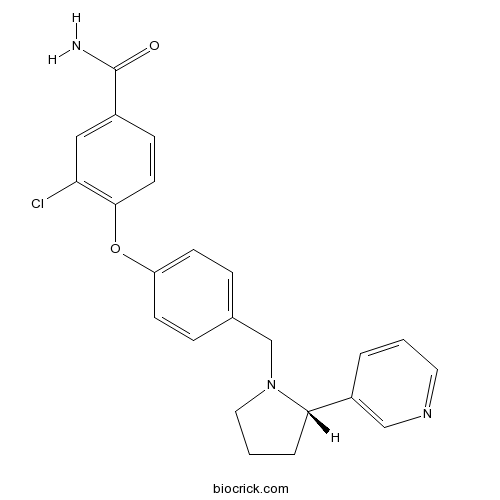
| Chemical Name | 3-chloro-4-[4-[[(2S)-2-pyridin-3-ylpyrrolidin-1-yl]methyl]phenoxy]benzamide | ||
| SMILES | C1CC(N(C1)CC2=CC=C(C=C2)OC3=C(C=C(C=C3)C(=O)N)Cl)C4=CN=CC=C4 | ||
| Standard InChIKey | LOOCZNLSXJHWTG-NRFANRHFSA-N | ||
| Standard InChI | InChI=1S/C23H22ClN3O2/c24-20-13-17(23(25)28)7-10-22(20)29-19-8-5-16(6-9-19)15-27-12-2-4-21(27)18-3-1-11-26-14-18/h1,3,5-11,13-14,21H,2,4,12,15H2,(H2,25,28)/t21-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

LY2795050 Dilution Calculator

LY2795050 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.4516 mL | 12.2582 mL | 24.5164 mL | 49.0328 mL | 61.291 mL |
| 5 mM | 0.4903 mL | 2.4516 mL | 4.9033 mL | 9.8066 mL | 12.2582 mL |
| 10 mM | 0.2452 mL | 1.2258 mL | 2.4516 mL | 4.9033 mL | 6.1291 mL |
| 50 mM | 0.049 mL | 0.2452 mL | 0.4903 mL | 0.9807 mL | 1.2258 mL |
| 100 mM | 0.0245 mL | 0.1226 mL | 0.2452 mL | 0.4903 mL | 0.6129 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Description: IC50 Value: 0.72 nM (κ-opioid Receptor); 25.8 nM (κ-opioid) [1] LY2795050 is a novel selective KOR antagonist and has the potential as a PET tracer to image KOR in vivo. in vitro: LY2795050 displays full antagonist activity and high binding affinity and selectivity for KOR with a ki value of 0.72 nM [1]. in vivo: In the brain, (11)C-LY2795050 displayed fast uptake kinetics (regional activity peak times of <20 min) and an uptake pattern consistent with the distribution of KOR in primates [1]. The LY2795050 ED50 at MOR was 119 μg/kg based on a 1-site model for 11C-carfentanil. The 1-site binding model was also deemed sufficient to describe the specific binding of 11C-LY2795050 at KOR. The ED50 at KOR estimated from the 1-site model was 15.6 μg/kg. Thus, the ED50 ratio for MOR:KOR was 7.6 [2]. Clinical trial: N/A
- ML 154
Catalog No.:BCC8022
CAS No.:1345964-89-7
- 5,7-Di-O-methylquercetin
Catalog No.:BCN3386
CAS No.:13459-07-9
- CYM 50308
Catalog No.:BCC6260
CAS No.:1345858-76-5
- Altiratinib
Catalog No.:BCC6385
CAS No.:1345847-93-9
- Arginase inhibitor 1
Catalog No.:BCC4034
CAS No.:1345808-25-4
- ETP-46464
Catalog No.:BCC3913
CAS No.:1345675-02-6
- AM095
Catalog No.:BCC1351
CAS No.:1345614-59-6
- Atorvastatin Calcium
Catalog No.:BCC2319
CAS No.:134523-03-8
- U 90042
Catalog No.:BCC7465
CAS No.:134516-99-7
- Gardenoin J
Catalog No.:BCN7666
CAS No.:1345109-46-7
- 1-Cinnamoyl-3-hydroxypyrrolidine
Catalog No.:BCN6497
CAS No.:1344876-77-2
- Trimethylvinylammonium(1+)
Catalog No.:BCN1820
CAS No.:13448-18-5
- Planchol E
Catalog No.:BCN6882
CAS No.:1346137-02-7
- SA 57
Catalog No.:BCC6280
CAS No.:1346169-63-8
- ML 218 hydrochloride
Catalog No.:BCC6207
CAS No.:1346233-68-8
- Stavudine sodium
Catalog No.:BCC4263
CAS No.:134624-73-0
- Eriocitrin
Catalog No.:BCN1208
CAS No.:13463-28-0
- Zinc Pyrithione
Catalog No.:BCC5008
CAS No.:13463-41-7
- ML 240
Catalog No.:BCC5604
CAS No.:1346527-98-7
- GSK503
Catalog No.:BCC6386
CAS No.:1346572-63-1
- GSK126
Catalog No.:BCC1604
CAS No.:1346574-57-9
- GSK621
Catalog No.:BCC6517
CAS No.:1346607-05-3
- GSK343
Catalog No.:BCC1607
CAS No.:1346704-33-3
- Lamivudine
Catalog No.:BCC3801
CAS No.:134678-17-4
Test-retest reproducibility of binding parameters in humans with 11C-LY2795050, an antagonist PET radiotracer for the kappa opioid receptor.[Pubmed:25593119]
J Nucl Med. 2015 Feb;56(2):243-8.
UNLABELLED: (11)C-LY2795050 is a new antagonist PET radioligand for the kappa opioid receptor (KOR). In this study, we assessed the reproducibility of the binding parameters of (11)C-LY2795050 in healthy human subjects. METHODS: Sixteen healthy subjects (11 men and 5 women) underwent 2 separate 90-min PET scans with arterial input function and plasma free fraction (fP) measurements. The 2-tissue-compartment model and multilinear analysis-1 were applied to calculate 5 outcome measures in 14 brain regions: distribution volume (VT), VT normalized by fP (VT/fP), and 3 binding potentials (nondisplaceable binding potential, binding potential relative to total plasma concentration, and binding potential relative to free plasma concentration: BPND, BPP, BPF, respectively). Since KOR is distributed ubiquitously throughout the brain, there are no suitable reference regions. We used a fixed fraction of individual cerebellar VT value (VT,CER) as the nondisplaceable VT (VND) (VND = VT,CER/1.17). The relative and absolute test-retest variability and intraclass correlation coefficient were evaluated for the outcome measures of (11)C-LY2795050. RESULTS: The test-retest variability of (11)C-LY2795050 for VT was no more than 10% in any region and was 12% in the amygdala. For binding potential (BPND and BPP), the test-retest variability was good in regions of moderate and high KOR density (BPND > 0.4) and poor in regions of low density. Correction by fP (VT/fP or BPF) did not improve the test-retest performance. CONCLUSION: Our results suggest that quantification of (11)C-LY2795050 imaging is reproducible and reliable in regions with moderate and high KOR density. Therefore, we conclude that this first antagonist radiotracer is highly useful for PET studies of KOR.
Kinetic modeling of (11)C-LY2795050, a novel antagonist radiotracer for PET imaging of the kappa opioid receptor in humans.[Pubmed:25182664]
J Cereb Blood Flow Metab. 2014 Nov;34(11):1818-25.
(11)C-LY2795050 is a novel kappa opioid receptor (KOR) antagonist tracer for positron emission tomography (PET) imaging. The purpose of this first-in-human study was to determine the optimal kinetic model for analysis of (11)C-LY2795050 imaging data. Sixteen subjects underwent baseline scans and blocking scans after oral naltrexone. Compartmental modeling and multilinear analysis-1 (MA1) were applied using the arterial input functions. Two-tissue compartment model and MA1 were found to be the best models to provide reliable measures of binding parameters. The rank order of (11)C-LY2795050 distribution volume (VT) matched the known regional KOR densities in the human brain. Blocking scans with naltrexone indicated no ideal reference region for (11)C-LY2795050. Three methods for calculation of the nondisplaceable distribution volume (VND) were assessed: (1) individual VND estimated from naltrexone occupancy plots, (2) mean VND across subjects, and (3) a fixed fraction of cerebellum VT. Approach (3) produced the lowest intersubject variability in the calculation of binding potentials (BPND, BPF, and BPP). Therefore, binding potentials of (11)C-LY2795050 can be determined if the specific binding fraction in the cerebellum is presumed to be unchanged by diseases and experimental conditions. In conclusion, results from the present study show the suitability of (11)C-LY2795050 to image and quantify KOR in humans.
Determination of the in vivo selectivity of a new kappa-opioid receptor antagonist PET tracer 11C-LY2795050 in the rhesus monkey.[Pubmed:23918735]
J Nucl Med. 2013 Sep;54(9):1668-74.
UNLABELLED: (11)C-LY2795050 is a novel kappa-selective antagonist PET tracer. The in vitro binding affinities (Ki) of LY2795050 at the kappa-opioid (KOR) and mu-opioid (MOR) receptors are 0.72 and 25.8 nM, respectively. Thus, the in vitro KOR/MOR binding selectivity is about 36:1. Our goal in this study was to determine the in vivo selectivity of this new KOR antagonist tracer in the monkey. METHODS: To estimate the ED50 value (dose of a compound [or drug] that gives 50% occupancy of the target receptor) of LY2795050 at the MOR and KOR sites, 2 series of blocking experiments were performed in 3 rhesus monkeys using (11)C-LY2795050 and (11)C-carfentanil with coinjections of various doses of unlabeled LY2795050. Kinetic modeling was applied to calculate regional binding potential (BP(ND)), and 1- and 2-site binding curves were fitted to these data to measure (11)C-LY2795050 binding selectivity. RESULTS: The LY2795050 ED50 at MOR was 119 mug/kg based on a 1-site model for (11)C-carfentanil. The 1-site binding model was also deemed sufficient to describe the specific binding of (11)C-LY2795050 at KOR. The ED50 at KOR estimated from the 1-site model was 15.6 mug/kg. Thus, the ED50 ratio for MOR:KOR was 7.6. CONCLUSION: The in vivo selectivity of (11)C-LY2795050 for KOR over MOR is 7.6. (11)C-LY2795050 has 4.7-fold-lower selectivity at KOR over MOR in vivo as compared with in vitro. Nevertheless, on the basis of our finding in vivo, 88% of the PET-observed specific binding of (11)C-LY2795050 under baseline conditions will be due to binding of the tracer at the KOR site in a region with similar prevalence of KOR and MOR. (11)C-LY2795050 is sufficiently selective for KOR over MOR in vivo to be considered an appropriate probe for studying the KOR with PET.
Receptor Occupancy of the kappa-Opioid Antagonist LY2456302 Measured with Positron Emission Tomography and the Novel Radiotracer 11C-LY2795050.[Pubmed:26628406]
J Pharmacol Exp Ther. 2016 Feb;356(2):260-6.
The kappa-opioid receptor (KOR) is thought to play an important therapeutic role in a wide range of neuropsychiatric and substance abuse disorders, including alcohol dependence. LY2456302 is a recently developed KOR antagonist with high affinity and selectivity and showed efficacy in the suppression of ethanol consumption in rats. This study investigated brain penetration and KOR target engagement after single oral doses (0.5-25 mg) of LY2456302 in 13 healthy human subjects. Three positron emission tomography scans with the KOR antagonist radiotracer (11)C-LY2795050 were conducted at baseline, 2.5 hours postdose, and 24 hours postdose. LY2456302 was well tolerated in all subjects without serious adverse events. Distribution volume was estimated using the multilinear analysis 1 method for each scan. Receptor occupancy (RO) was derived from a graphical occupancy plot and related to LY2456302 plasma concentration to determine maximum occupancy (rmax) and IC50. LY2456302 dose dependently blocked the binding of (11)C-LY2795050 and nearly saturated the receptors at 10 mg, 2.5 hours postdose. Thus, a dose of 10 mg of LY2456302 appears well suited for further clinical testing. Based on the pharmacokinetic (PK)-RO model, the rmax and IC50 of LY2456302 were estimated as 93% and 0.58 ng/ml to 0.65 ng/ml, respectively. Assuming that rmax is 100%, IC50 was estimated as 0.83 ng/ml.


