PF-04971729SGLT inhibitor CAS# 1210344-57-2 |
- LX-4211
Catalog No.:BCC1714
CAS No.:1018899-04-1
- Dapagliflozin
Catalog No.:BCC2552
CAS No.:461432-26-8
- Canagliflozin
Catalog No.:BCC3696
CAS No.:842133-18-0
- Empagliflozin (BI 10773)
Catalog No.:BCC2472
CAS No.:864070-44-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1210344-57-2 | SDF | Download SDF |
| PubChem ID | 44814423 | Appearance | Powder |
| Formula | C22H25ClO7 | M.Wt | 436.88 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Ertugliflozin | ||
| Solubility | DMSO : ≥ 46 mg/mL (105.29 mM) *"≥" means soluble, but saturation unknown. | ||
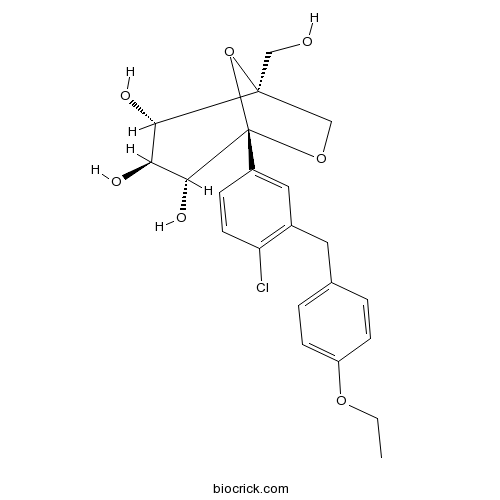
| Chemical Name | (1S,2S,3S,4R,5S)-5-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol | ||
| SMILES | CCOC1=CC=C(C=C1)CC2=C(C=CC(=C2)C34C(C(C(C(O3)(CO4)CO)O)O)O)Cl | ||
| Standard InChIKey | MCIACXAZCBVDEE-CUUWFGFTSA-N | ||
| Standard InChI | InChI=1S/C22H25ClO7/c1-2-28-16-6-3-13(4-7-16)9-14-10-15(5-8-17(14)23)22-20(27)18(25)19(26)21(11-24,30-22)12-29-22/h3-8,10,18-20,24-27H,2,9,11-12H2,1H3/t18-,19-,20+,21-,22-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

PF-04971729 Dilution Calculator

PF-04971729 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.289 mL | 11.4448 mL | 22.8896 mL | 45.7792 mL | 57.224 mL |
| 5 mM | 0.4578 mL | 2.289 mL | 4.5779 mL | 9.1558 mL | 11.4448 mL |
| 10 mM | 0.2289 mL | 1.1445 mL | 2.289 mL | 4.5779 mL | 5.7224 mL |
| 50 mM | 0.0458 mL | 0.2289 mL | 0.4578 mL | 0.9156 mL | 1.1445 mL |
| 100 mM | 0.0229 mL | 0.1144 mL | 0.2289 mL | 0.4578 mL | 0.5722 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
PF-04971729, a potent and selective inhibitor of the sodium-dependent glucose cotransporter 2, is currently in phase 2 trials for the treatment of diabetes mellitus. Inhibitory effects against the organic cation transporter 2-mediated uptake of [14C]metformin by PF-04971729 also were very weak (IC50 900μM).The disposition of PF-04971729, an orally active selective inhibitor of the sodium-dependent glucose cotransporter 2, was studied after a single 25-mg oral dose of [14C]-PF-04971729 to healthy human subjects. The absorption of PF-04971729 in humans was rapid with a Tmax at ~ 1.0 h. Of the total radioactivity excreted in feces and urine, unchanged PF-04971729 collectively accounted for ~ 35.3% of the dose, suggestive of moderate metabolic elimination in humans.
- IEM 1460
Catalog No.:BCC7135
CAS No.:121034-89-7
- Nelarabine
Catalog No.:BCC1072
CAS No.:121032-29-9
- Secretin (rat)
Catalog No.:BCC5848
CAS No.:121028-49-7
- JZL 195
Catalog No.:BCC7966
CAS No.:1210004-12-8
- N-Acetyl-5-Hydroxytryptamine
Catalog No.:BCC9080
CAS No.:1210-83-9
- ST 1936 oxalate
Catalog No.:BCC7919
CAS No.:1210-81-7
- 3'-Nitroacetophenone
Catalog No.:BCN2256
CAS No.:121-89-1
- Propyl gallate
Catalog No.:BCN8431
CAS No.:121-79-9
- 2-Amino-5-nitrothiazole
Catalog No.:BCC8538
CAS No.:121-66-4
- N-Acetylsulfanilyl chloride
Catalog No.:BCC9084
CAS No.:121-60-8
- Benzethonium Chloride
Catalog No.:BCC4635
CAS No.:121-54-0
- (-)-Terreic acid
Catalog No.:BCC7051
CAS No.:121-40-4
- Abiesadine I
Catalog No.:BCN6104
CAS No.:1210347-50-4
- Melanotan II
Catalog No.:BCC7414
CAS No.:121062-08-6
- 3-O-trans-p-Coumaroyltormentic acid
Catalog No.:BCN4724
CAS No.:121064-78-6
- 3-O-cis-p-Coumaroyltormentic acid
Catalog No.:BCN3184
CAS No.:121072-40-0
- L-670,596
Catalog No.:BCC5857
CAS No.:121083-05-4
- EG00229
Catalog No.:BCC5376
CAS No.:1210945-69-9
- Cefprozil hydrate
Catalog No.:BCC4951
CAS No.:121123-17-9
- LEE011
Catalog No.:BCC3926
CAS No.:1211441-98-3
- LEE011 hydrochloride
Catalog No.:BCC4101
CAS No.:1211443-80-9
- Rauvoyunine C
Catalog No.:BCN4833
CAS No.:1211543-01-9
- TC-N 1752
Catalog No.:BCC6179
CAS No.:1211866-85-1
- [Ala1,3,11,15]-Endothelin
Catalog No.:BCC5731
CAS No.:121204-87-3
Pharmacokinetics, metabolism, and excretion of the antidiabetic agent ertugliflozin (PF-04971729) in healthy male subjects.[Pubmed:23169609]
Drug Metab Dispos. 2013 Feb;41(2):445-56.
The disposition of ertugliflozin (PF-04971729), an orally active selective inhibitor of the sodium-dependent glucose cotransporter 2, was studied after a single 25-mg oral dose of [(14)C]-ertugliflozin to healthy human subjects. Mass balance was achieved with approximately 91% of the administered dose recovered in urine and feces. The total administered radioactivity excreted in feces and urine was 40.9% and 50.2%, respectively. The absorption of ertugliflozin in humans was rapid with a T(max) at approximately 1.0 hour. Of the total radioactivity excreted in feces and urine, unchanged ertugliflozin collectively accounted for approximately 35.3% of the dose, suggestive of moderate metabolic elimination in humans. The principal biotransformation pathway involved glucuronidation of the glycoside hydroxyl groups to yield three regioisomeric metabolites, M4a, M4b, and M4c ( approximately 39.3% of the dose in urine), of which M4c was the major regioisomer ( approximately 31.7% of the dose). The structure of M4a and M4c were confirmed to be ertugliflozin -4-O-beta- and -3-O-beta-glucuronide, respectively, via comparison of the HPLC retention time and mass spectra with authentic standards. A minor metabolic fate involved oxidation by cytochrome P450 to yield monohydroxylated metabolites M1 and M3 and des-ethyl ertugliflozin (M2), which accounted for approximately 5.2% of the dose in excreta. In plasma, unchanged ertugliflozin and the corresponding 4-O-beta- (M4a) and 3-O-beta- (M4c) glucuronides were the principal components, which accounted for 49.9, 12.2, and 24.1% of the circulating radioactivity. Overall, these data suggest that ertugliflozin is well absorbed in humans, and eliminated largely via glucuronidation.
Preclinical species and human disposition of PF-04971729, a selective inhibitor of the sodium-dependent glucose cotransporter 2 and clinical candidate for the treatment of type 2 diabetes mellitus.[Pubmed:21690265]
Drug Metab Dispos. 2011 Sep;39(9):1609-19.
(1S,2S,3S,4R,5S)-5-[4-Chloro-3-(4-ethoxybenzyl)phenyl]-1-hydroxymethyl-6,8-dioxab icyclo[3.2.1]octane-2,3,4-triol (PF-04971729), a potent and selective inhibitor of the sodium-dependent glucose cotransporter 2, is currently in phase 2 trials for the treatment of diabetes mellitus. This article describes the preclinical species and in vitro human disposition characteristics of PF-04971729 that were used in experiments performed to support the first-in-human study. Plasma clearance was low in rats (4.04 ml . min(-1) . kg(-1)) and dogs (1.64 ml . min(-1) . kg(-1)), resulting in half-lives of 4.10 and 7.63 h, respectively. Moderate to good bioavailability in rats (69%) and dogs (94%) was observed after oral dosing. The in vitro biotransformation profile of PF-04971729 in liver microsomes and cryopreserved hepatocytes from rat, dog, and human was qualitatively similar; prominent metabolic pathways included monohydroxylation, O-deethylation, and glucuronidation. No human-specific metabolites of PF-04971729 were detected in in vitro studies. Reaction phenotyping studies using recombinant enzymes indicated a role of CYP3A4/3A5, CYP2D6, and UGT1A9/2B7 in the metabolism of PF-04971729. No competitive or time-dependent inhibition of the major human cytochrome P450 enzymes was discerned with PF-04971729. Inhibitory effects against the organic cation transporter 2-mediated uptake of [(14)C]metformin by PF-04971729 also were very weak (IC(50) = approximately 900 muM). Single-species allometric scaling of rat pharmacokinetics of PF-04971729 was used to predict human clearance, distribution volume, and oral bioavailability. Human pharmacokinetic predictions were consistent with the potential for a low daily dose. First-in-human studies after oral administration indicated that the human pharmacokinetics/dose predictions for PF-04971729 were in the range that is likely to yield a favorable pharmacodynamic response.


