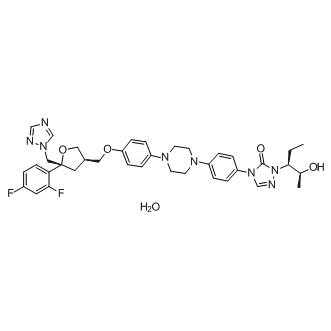
Posaconazole hydrateC14ɑ demethylase inhibitor CAS# 1198769-38-8 |
- DCC-2036 (Rebastinib)
Catalog No.:BCC4390
CAS No.:1020172-07-9
- Imatinib Mesylate (STI571)
Catalog No.:BCC1115
CAS No.:220127-57-1
- PD173955
Catalog No.:BCC3999
CAS No.:260415-63-2
- Nilotinib(AMN-107)
Catalog No.:BCC3643
CAS No.:641571-10-0
- GNF 5
Catalog No.:BCC3892
CAS No.:778277-15-9
- Bafetinib (INNO-406)
Catalog No.:BCC4036
CAS No.:887650-05-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1198769-38-8 | SDF | Download SDF |
| PubChem ID | 78357825 | Appearance | Powder |
| Formula | C37H44F2N8O5 | M.Wt | 718.79 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in DMSO | ||
| Chemical Name | 4-[4-[4-[4-[[(3R,5R)-5-(2,4-difluorophenyl)-5-(1,2,4-triazol-1-ylmethyl)oxolan-3-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2-[(2S,3S)-2-hydroxypentan-3-yl]-1,2,4-triazol-3-one;hydrate | ||
| SMILES | CCC(C(C)O)N1C(=O)N(C=N1)C2=CC=C(C=C2)N3CCN(CC3)C4=CC=C(C=C4)OCC5CC(OC5)(CN6C=NC=N6)C7=C(C=C(C=C7)F)F.O | ||
| Standard InChIKey | TUNUVJALSUOKOC-VPJFHDDISA-N | ||
| Standard InChI | InChI=1S/C37H42F2N8O4.H2O/c1-3-35(26(2)48)47-36(49)46(25-42-47)31-7-5-29(6-8-31)43-14-16-44(17-15-43)30-9-11-32(12-10-30)50-20-27-19-37(51-21-27,22-45-24-40-23-41-45)33-13-4-28(38)18-34(33)39;/h4-13,18,23-27,35,48H,3,14-17,19-22H2,1-2H3;1H2/t26-,27+,35-,37-;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Posaconazole hydrate Dilution Calculator

Posaconazole hydrate Molarity Calculator

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Posaconazole is a broad-spectrum, second generation, triazole compound with antifungal activity. Posaconazole strongly inhibits 14-alpha demethylase, a cytochrome P450-dependent enzyme. Posaconazole is a sterol C14ɑ demethylase inhibitor with an IC50 of 0.25 nM. Inhibition of 14-alpha-demethylase prevents the conversion of lanosterol to ergosterol, an important component of the fungal cell wall. Inhibition of ergosterol synthesis changes the fungal cell membrane composition and integrity, alters membrane permeability and eventually leads to fungal cell lysis. Compared to other azole antifungals, posaconazole is a significantly more potent inhibitor of sterol 14-alpha demethylase. Posaconazole is the most advanced candidate for the treatment of Chagas disease. Posaconazole has entered in a phase II clinical trial in the treatment of mycoses.
- Esomeprazole magnesium salt
Catalog No.:BCC5430
CAS No.:1198768-91-0
- Phyperunolide E
Catalog No.:BCN7292
CAS No.:1198400-52-0
- Coptisine sulfate
Catalog No.:BCN2286
CAS No.:1198398-71-8
- Cerdulatinib (PRT062070)
Catalog No.:BCC8068
CAS No.:1198300-79-6
- Sodium Channel inhibitor 1
Catalog No.:BCC1959
CAS No.:1198117-23-5
- Mirin
Catalog No.:BCC5986
CAS No.:1198097-97-0
- AP26113
Catalog No.:BCC1069
CAS No.:1197958-12-5
- 3-Furfuryl 2-pyrrolecarboxylate
Catalog No.:BCN6086
CAS No.:119767-00-9
- 29-Norlanosta-8,24-diene-1alpha,2alpha,3beta-triol
Catalog No.:BCN7984
CAS No.:119765-92-3
- Baohuoside VI
Catalog No.:BCC8129
CAS No.:119760-73-5
- Tautomycetin
Catalog No.:BCC7320
CAS No.:119757-73-2
- Schizanthine M
Catalog No.:BCN1939
CAS No.:119736-78-6
- CGRP 8-37 (human)
Catalog No.:BCC5724
CAS No.:119911-68-1
- 7-O-Methylmorroniside
Catalog No.:BCN7293
CAS No.:119943-46-3
- Peroxy Orange 1
Catalog No.:BCC6336
CAS No.:1199576-10-7
- 1,2-Didehydrotanshinone IIA
Catalog No.:BCN3143
CAS No.:119963-50-7
- INT-777
Catalog No.:BCC5390
CAS No.:1199796-29-6
- Unedone
Catalog No.:BCN6759
CAS No.:1199815-09-2
- Sulfuretin
Catalog No.:BCN4725
CAS No.:120-05-8
- Scoparone
Catalog No.:BCN6088
CAS No.:120-08-1
- Veratraldehyde
Catalog No.:BCN6089
CAS No.:120-14-9
- Tropine benzilate
Catalog No.:BCN1921
CAS No.:3736-36-5
- Clorofene
Catalog No.:BCC8919
CAS No.:120-32-1
- 3-Amino-4-methoxybenzanilide
Catalog No.:BCC8613
CAS No.:120-35-4
Gateways to clinical trials.[Pubmed:17136234]
Methods Find Exp Clin Pharmacol. 2006 Oct;28(8):533-91.
Gateways to Clinical Trials are a guide to the most recent clinical trials in current literature and congresses. The data the following tables have been retrieved from the Clinical Trials Knowledge Area of Prous Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issues focuses on the following selection of drugs: (-)-Epigallocatechin gallate, (-)-gossypol, 2-deoxyglucose, 3,4-DAP, 7-monohydroxyethylrutoside; Ad5CMV-p53, adalimumab, adefovir dipivoxil, ADH-1, alemtuzumab, aliskiren fumarate, alvocidib hydrochloride, aminolevulinic acid hydrochloride, aminolevulinic acid methyl ester, amrubicin hydrochloride, AN-152, anakinra, anecortave acetate, antiasthma herbal medicine intervention, AP-12009, AP-23573, apaziquone, aprinocarsen sodium, AR-C126532, AR-H065522, aripiprazole, armodafinil, arzoxifene hydrochloride, atazanavir sulfate, atilmotin, atomoxetine hydrochloride, atorvastatin, avanafil, azimilide hydrochloride; Bevacizumab, biphasic insulin aspart, BMS-214662, BN-83495, bortezomib, bosentan, botulinum toxin type B; Caspofungin acetate, cetuximab, chrysin, ciclesonide, clevudine, clofarabine, clopidogrel, CNF-1010, CNTO-328, CP-751871, CX-717, Cypher; Dapoxetine hydrochloride, darifenacin hydrobromide, dasatinib, deferasirox, dextofisopam, dextromethorphan/quinidine sulfate, diclofenac, dronedarone hydrochloride, drotrecogin alfa (activated), duloxetine hydrochloride, dutasteride; Edaravone, efaproxiral sodium, emtricitabine, entecavir, eplerenone, epratuzumab, erlotinib hydrochloride, escitalopram oxalate, etoricoxib, ezetimibe, ezetimibe/simvastatin; Finrozole, fipamezole hydrochloride, fondaparinux sodium, fulvestrant; Gabapentin enacarbil, gaboxadol, gefitinib, gestodene, ghrelin (human); Human insulin, human papillomavirus vaccine; Imatinib mesylate, immunoglobulin intravenous (human), indiplon, insulin detemir, insulin glargine, insulin glulisine, intranasal insulin, istradefylline, i.v. gamma-globulin, ivabradine hydrochloride, ixabepilone; LA-419, lacosamide, landiolol, lanthanum carbonate, lidocaine/prilocaine, liposomal cisplatin, lutropin alfa; Matuzumab, MBP(82-98), mecasermin, MGCD-0103, MMR-V, morphine hydrochloride, mycophenolic acid sodium salt; Natalizumab, NCX-4016, neridronic acid, nesiritide, nilotinib, NSC-330507; O6-benzylguanine, olanzapine/fluoxetine hydrochloride, omalizumab; Panitumumab, parathyroid hormone (human recombinant), parecoxib sodium, PEG-filgrastim, peginterferon alfa-2a, peginterferon alfa-2b, pegvisomant, pemetrexed disodium, perospirone hydrochloride, pexelizumab, phorbol 12-myristate 13-acetate, pneumococcal 7-valent conjugate vaccine, posaconazole, pramiconazole, prasugrel, pregabalin, prilocaine; rAAV-GAD65, raclopride, rasagiline mesilate, retapamulin, rosuvastatin calcium, rotigotine, rufinamide; SarCNU, SB-743921, SHL-749, sirolimus-eluting stent, sitaxsentan sodium, sorafenib; TachoSil, tadalafil, talampanel, Taxus, tegaserod maleate, telithromycin, telmisartan/hydrochlorothiazide, temsirolimus, tenatoprazole, teriflunomide, tetrathiomolybdate, ticilimumab, timcodar dimesilate, tipifarnib, tirapazamine, TPI, tramiprosate, trifluridine/TPI, trimethoprim; Ularitide, Urocortin 2; Valdecoxib, valganciclovir hydrochloride, valproate magnesium, valspodar, vardenafil hydrochloride hydrate, vitespen, vofopitant hydrochloride, volociximab, vorinostat; Yttrium 90 (90Y) ibritumomab tiuxetan; Ziprasidone hydrochloride, zotarolimus, zotarolimus-eluting stent.
Gateways to clinical trials.[Pubmed:19536362]
Methods Find Exp Clin Pharmacol. 2009 Apr;31(3):183-226.
(+)-Dapoxetine hydrochloride, [(123)I]-BZA, 9-Aminocamptothecin; Abacavir sulfate/lamivudine, Adalimumab, Adefovir dipivoxil, Alemtuzumab, Alvocidib hydrochloride, Ambrisentan, Amsilarotene, Anacetrapib, Anakinra, Apricitabine, Aripiprazole, Arsenic trioxide, Atazanavir sulfate, Atazanavir/ritonavir, Atrasentan, Azacitidine; Banoxantrone, Bazedoxifene acetate, Bevacizumab, Bexarotene, Biphasic insulin aspart, Bortezomib, Bosentan, Bromfenac; Cachectin, Calcipotriol/betamethasone dipropionate, Canakinumab, Carfilzomib, CAT-354, CCX-282, Certolizumab pegol, Cetuximab, Choline fenofibrate, Clevudine, Clofarabine, CNTO-328, Corifollitropin alfa, Crofelemer; Daptomycin, Darbepoetin alfa, Darunavir, Dasatinib, Decitabine, Deferasirox, Denosumab, Duloxetine hydrochloride, Dutasteride; Emtricitabine, Enfuvirtide, Entecavir, Epoetin zeta, Erlotinib hydrochloride, Escitalopram oxalate, Eslicarbazepine acetate, Eszopiclone, Etravirine, Everolimus, Exenatide, Ezetimibe, Ezetimibe/simvastatin; Farglitazar, Febuxostat, Fosamprenavir calcium, FX-06; Gabapentin enacarbil, Gefitinib; HIVIS DNA; Imatinib mesylate, INCB- 18424, Indacaterol, Inotuzumab ozogamicin, Insulin detemir; JNJ-26854165; Lacosamide, Landiolol, Laromustine, Lenalidomide, Liposomal doxorubicin, L-NAME, Lopinavir, Lopinavir/ritonavir, Lumiracoxib; Maraviroc, Mepolizumab, Methoxy polyethylene glycol- epoetin-beta, Miglustat, MK-0493, MVA-CMDR, Mycophenolic acid sodium salt; Natalizumab, Nepafenac, Neratinib, Neridronic acid, Nesiritide, Nilotinib hydrochloride monohydrate; Olmesartan medoxomil, Omacetaxine mepesuccinate, Omalizumab; Paclitaxel poliglumex, Palifermin, Patupilone, Pegfilgrastim, Peginterferon alfa-2a, Peginterferon alfa-2b, Peginterferon alfa-2b/ ribavirin, Pemetrexed disodium, PHA-848125, Pitavastatin calcium, Posaconazole, Povidone-iodine liposome complex, Prasugrel, Pregabalin, Prucalopride; Raltegravir potassium, Retigabine, Revaprazan hydrochloride, rhFSH, Rilpivirine, Rivaroxaban, Romidepsin, Rosuvastatin calcium, RWJ-676070; SAR-109659, Sitagliptin phosphate monohydrate, Sorafenib, Stavudine/Lamivudine/Nevirapine, Sunitinib malate; Tadalafil, Telaprevir, Telbivudine, Tenofovir disoproxil fumarate, Tenofovir disoproxil fumarate/emtricitabine, Tenofovir disoproxil fumarate/emtricitabine/efavirenz, Teriparatide, Tigecycline, Tiotropium bromide, Tipifarnib, Tipranavir, Tocilizumab, Trifluridine/TPI; UP-780; Vandetanib, Vardenafil hydrochloride hydrate, Vatalanib succinate, Vitespen, Vorinostat; Yttrium 90 (90Y) ibritumomab tiuxetan; Zoledronic acid monohydrate.
Gateways to clinical trials.[Pubmed:18389098]
Methods Find Exp Clin Pharmacol. 2008 Jan-Feb;30(1):67-99.
Gateways to Clinical Trials are a guide to the most recent clinical trials in current literature and congresses. The data in the following tables has been retrieved from the Clinical Trials Knowledge Area of Prouse Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issue focuses on the following selection of drugs: 101M, 3F8; Abatacept, ABT-263, Adalimumab, AG-7352, Agatolimod sodium, Alfimeprase, Aliskiren fumarate, Alvimopan hydrate, Aminolevulinic acid hexyl ester, Ammonium tetrathiomolybdate, Anakinra, Aripiprazole, AS-1404, AT-9283, Atomoxetine hydrochloride, AVE-1642, AVE-9633, Axitinib, AZD-0530; Becocalcidiol, Belotecan hydrochloride, Bevacizumab, BG-9928, BIBF-1120, BMS-275183, Bortezomib, Bosentan; Catumaxomab, Cetuximab, CHR-2797, Ciclesonide, Clevidipine, Cypher, Cytarabine/daunorubicin; Darifenacin hydrobromide, Darunavir, Denosumab, Desvenlafaxine succinate, Disufenton sodium, Duloxetine hydrochloride, Dutasteride; Eculizumab, Efalizumab, Eicosapentaenoic acid/docosahexaenoic acid, Eplerenone, Epratuzumab, Erlotinib hydrochloride, Escitalopram oxalate, Ethynylcytidine, Etravirine, Everolimus, Ezetimibe; Fulvestrant; Garenoxacin mesilate, Gefitinib, Gestodene; HI-164, Hydralazine hydrochloride/isosorbide dinitrate; Icatibant acetate, ICX-RHY, Idraparinux sodium, Indacaterol, Ispronicline, Ivabradine hydrochloride, Ixabepilone; KB-2115, KW-2449; L-791515, Lapatinib ditosylate, LGD-4665, Licofelone, Liposomal doxorubicin, Lisdexamfetamine mesilate, Lumiracoxib; Methoxy polyethylene glycol-epoetin-beta, Miglustat, Mipomersen sodium, Mitumprotimut-T, MK-0822A, MK-0974; Nelarabine; Obatoclax mesylate, Olmesartan medoxomil, Olmesartan medoxomil/hydrochlorothiazide; Paliperidone, Palonosetron hydrochloride, Panitumumab, Pegfilgrastim, Peginterferon alfa-2a, Pemetrexed disodium, Perospirone hydrochloride, Pertuzumab, Pimecrolimus, Pitrakinra, Pixantrone maleate, Posaconazole, Pregabalin; Quercetin; RALGA, Raltegravir potassium, Ranelic acid distrontium salt, rhHistone 1.3, Rimonabant, Rivaroxaban, Rosuvastatin calcium, RTS,S/SBAS2; Satraplatin, SNDX-275, Sodium butyrate, Solifenacin succinate, Sorafenib, SU-14813, Sunitinib malate; Tadalafil, Tafenoquine succinate, Tamatinib fosdium, Taxus, Telbivudine, Telmisartan/hydrochlorothiazide, Temsirolimus, Tiotropium bromide, Tipranavir, Tocilizumab, Trabectedin, Tramadol hydrochloride/acetaminophen; Ulipristal acetate, Uracil, Ursodeoxycholyltaurine; Valdecoxib, Vardenafil hydrochloride hydrate, Varenicline tartrate, Vildagliptin, Vinflunine, Vitespen, Vorinostat; ZK-EPO, Zoledronic acid monohydrate.
Gateways to clinical trials.[Pubmed:18040531]
Methods Find Exp Clin Pharmacol. 2007 Oct;29(8):547-83.
(-)-Epigallocatechin gallate, [188Re]-P2045, 12B75, 89-12; Abacavir sulfate/lamivudine, Abatacept, Abiraterone acetate, ABT-869, Adalimumab, Ad-rh Endostatin, AI-700, Alemtuzumab, Alvimopan hydrate, Amrubicin hydrochloride, AP-12009, Apomab 7.3, Arformoterol tartrate, Aripiprazole, AS-1404, Azacitidine, AZD-0530; Bevacizumab, BHT-3009, Biapenem, Bortezomib, Bosentan, Bremelanotide; CA9-SCAN, Calcitonin gene-related peptide, Canertinib dihydrochloride, Cannabidiol, Carboxyamidotriazole, Caspofungin acetate, Celgosivir, Certolizumab pegol, Cinacalcet hydrochloride, Clevudine, CP-751871, Curcumin, Cx-401, Cypher; Darunavir, Decitabine, Deforolimus, Dexamet, Dipyridamole/prednisolone, Drospirenone, Drospirenone/estradiol, DTPw-HepB-Hib, Duloxetine hydrochloride; Efalizumab, Emtricitabine, Erlotinib hydrochloride, Escitalopram oxalate, Eszopiclone; Ferumoxtran-10, Ferumoxytol, Fondaparinux sodium, Fosaprepitant dimeglumine; gamma-Hydroxybutyrate sodium, Gefitinib, Genistein, Ghrelin (human), Gimatecan, GM-CSF PMED, Golimumab, gp100 PMED; Imatinib mesylate, Immunoglobulin intravenous (human), IV Gamma-globulin; LA-419, Laropiprant, L-BLP-25, Levodopa/carbidopa/entacapone, Lidocaine/prilocaine, Lopinavir/ritonavir, Lumiracoxib, LY-2076962; Mepolizumab, Methylnaltrexone bromide, Mitiglinide calcium hydrate, Mycophenolic acid sodium salt, Myristyl nicotinate; Natalizumab, Nesiritide, Niacin/lovastatin; Oblimersen sodium, Ofatumumab, Olmesartan medoxomil, Olmesartan medoxomil/hydrochlorothiazide, Ozarelix; Palonosetron hydrochloride, Parathyroid hormone (human recombinant), Pazopanib hydrochloride, Pegaptanib octasodium, Pegfilgrastim, Peginterferon alfa- 2a, Peginterferon alfa-2b, Pegvisomant, Pemetrexed disodium, Pexelizumab, Picoplatin, Pimecrolimus, Posaconazole, Pregabalin, PRO-1762, Progesterone caproate, Prulifloxacin; Ramelteon, Ranelic acid distrontium salt, Reparixin, Rosuvastatin calcium; Rotigotine; Satraplatin, Sertraline, Sipuleucel-T, SLIT-cisplatin, SNDX-275, Solifenacin succinate, Sunitinib malate; Tadalafil, Talnetant, Tanespimycin, Taxus, Tegaserod maleate, Telmisartan/hydrochlorothiazide, Tenofovir disoproxil fumarate/emtricitabine, Teriparatide, tgAAC-94, Tiotropium bromide, Tocilizumab, Tolvaptan, Trimethoprim; Vardenafil hydrochloride hydrate, Vatalanib succinate, Vinflunine, Voriconazole, VX-680; XL-880; Yttrium 90 (90Y) ibritumomab tiuxetan.
Gateways to clinical trials.[Pubmed:21225012]
Methods Find Exp Clin Pharmacol. 2010 Dec;32(10):749-73.
Gateways to Clinical Trials is a guide to the most recent clinical trials in current literature and congresses. The data in the following tables has been retrieved from the Clinical Trials Knowledge Area of Thomson Reuters Integrity(SM), the drug discovery and development portal, http://www.thomsonreutersintegrity.com. This issue focuses on the following selection of drugs: 17-Hydroxyprogesterone caproate; Abacavir sulfate/lamivudine, Aclidinium bromide, Adalimumab, Adefovir, Alemtuzumab, Alkaline phosphatase, Amlodipine, Apilimod mesylate, Aripiprazole, Axitinib, Azacitidine; Belotecan hydrochloride, Berberine iodide, Bevacizumab, Bortezomib, Bosentan, Bryostatin 1; Calcipotriol/hydrocortisone, Carglumic acid, Certolizumab pegol, Cetuximab, Cinacalcet hydrochloride, Cixutumumab, Coumarin, Custirsen sodium; Darbepoetin alfa, Darifenacin hydrobromide, Darunavir, Dasatinib, Denibulin hydrochloride, Denosumab, Diacetylmorphine, Dulanermin, Duloxetine hydrochloride; Ecogramostim, Enfuvirtide, Entecavir, Enzastaurin hydrochloride, Eplerenone, Escitalopram oxalate, Esomeprazole sodium, Etravirine, Everolimus, Ezetimibe; Fenofibrate/pravastatin sodium, Ferric carboxymaltose, Flavangenol, Fondaparinux sodium; Glutamine, GSK-1024850A; Hepatitis B hyperimmunoglobulin, Hib-MenC, HIV-LIPO-5; Immunoglobulin intravenous (human), Indacaterol maleate, Indibulin, Indium 111 ((1)(1)(1)In) ibritumomab tiuxetan, Influenza A (H1N1) 2009 Monovalent vaccine, Inhalable human insulin, Insulin glulisine; Lapatinib ditosylate, Leucovorin/UFT; Maraviroc, Mecasermin, MMR-V, Morphine hydrochloride, Morphine sulfate/naltrexone hydrochloride, Mycophenolic acid sodium salt; Naproxen/esomeprazole magnesium, Natalizumab; Oncolytic HSV; Paliperidone, PAN-811, Paroxetine, Pegfilgrastim, Peginterferon alfa-2a, Peginterferon alfa-2b/ribavirin, Pegvisomant, Pemetrexed disodium, Pimecrolimus, Posaconazole, Pregabalin; Raltegravir potassium, Ranelic acid distrontium salt, Rasburicase, Rilpivirine hydrochloride; Sertindole, Sivelestat sodium hydrate, Sorafenib, Sumatriptan succinate/naproxen sodium, Sunitinib malate; Tafluprost, Telithromycin, Temsirolimus, Tenofovir disoproxil fumavate, Tenofovir disoproxil fumarate/emtricitabine, Teriparatide, Ticagrelor, Tigecycline, Tipranavir, Tirapazamine, Trimetrexate; Ulipristal acetate; Valganciclovir hydrochloride, Vicriviroc, Vorinostat; Yttrium 90 (90Y) ibritumomab tiuxetan.


