3'-Fluorobenzylspiperone maleateD2-like receptor ligand CAS# 1135278-61-3 |
- (24R)-MC 976
Catalog No.:BCC1289
CAS No.:112828-09-8
- (24S)-MC 976
Catalog No.:BCC1291
CAS No.:112849-14-6
- 1alpha, 25-Dihydroxy VD2-D6
Catalog No.:BCC1299
CAS No.:216244-04-1
- 1alpha, 24, 25-Trihydroxy VD2
Catalog No.:BCC1298
CAS No.:457048-34-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1135278-61-3 | SDF | Download SDF |
| PubChem ID | 24978532 | Appearance | Powder |
| Formula | C34H35F2N3O6 | M.Wt | 619.67 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in DMSO > 10 mM | ||
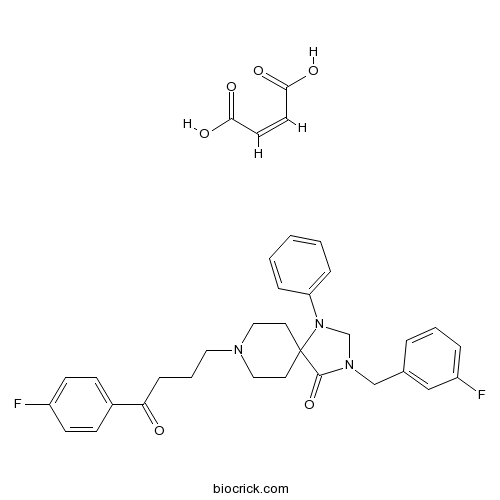
| Chemical Name | (Z)-but-2-enedioic acid;3-[(3-fluorophenyl)methyl]-8-[4-(4-fluorophenyl)-4-oxobutyl]-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one | ||
| SMILES | C1CN(CCC12C(=O)N(CN2C3=CC=CC=C3)CC4=CC(=CC=C4)F)CCCC(=O)C5=CC=C(C=C5)F.C(=CC(=O)O)C(=O)O | ||
| Standard InChIKey | JURUFADWVGXLOY-BTJKTKAUSA-N | ||
| Standard InChI | InChI=1S/C30H31F2N3O2.C4H4O4/c31-25-13-11-24(12-14-25)28(36)10-5-17-33-18-15-30(16-19-33)29(37)34(21-23-6-4-7-26(32)20-23)22-35(30)27-8-2-1-3-9-27;5-3(6)1-2-4(7)8/h1-4,6-9,11-14,20H,5,10,15-19,21-22H2;1-2H,(H,5,6)(H,7,8)/b;2-1- | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | A very potent and selective ligand for the D2 receptor (Ki = 0.023 nM). Compared with spiperone, displays a 2.5-fold greater affinity at D2 and a 12-fold lower affinity at 5-HT2 receptors. |

3'-Fluorobenzylspiperone maleate Dilution Calculator

3'-Fluorobenzylspiperone maleate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.6138 mL | 8.0688 mL | 16.1376 mL | 32.2752 mL | 40.3441 mL |
| 5 mM | 0.3228 mL | 1.6138 mL | 3.2275 mL | 6.455 mL | 8.0688 mL |
| 10 mM | 0.1614 mL | 0.8069 mL | 1.6138 mL | 3.2275 mL | 4.0344 mL |
| 50 mM | 0.0323 mL | 0.1614 mL | 0.3228 mL | 0.6455 mL | 0.8069 mL |
| 100 mM | 0.0161 mL | 0.0807 mL | 0.1614 mL | 0.3228 mL | 0.4034 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- CGP 78608 hydrochloride
Catalog No.:BCC7087
CAS No.:1135278-54-4
- SR 59230A hydrochloride
Catalog No.:BCC7094
CAS No.:1135278-41-9
- VU 0255035
Catalog No.:BCC7766
CAS No.:1135243-19-4
- VU 0357017 hydrochloride
Catalog No.:BCC7907
CAS No.:1135242-13-5
- Tracazolate hydrochloride
Catalog No.:BCC7115
CAS No.:1135210-68-2
- Moxidectin
Catalog No.:BCC5309
CAS No.:113507-06-5
- Ferulic acid
Catalog No.:BCN5948
CAS No.:1135-24-6
- Ivacaftor benzenesulfonate
Catalog No.:BCC1663
CAS No.:1134822-09-5
- Ivacaftor hydrate
Catalog No.:BCC1664
CAS No.:1134822-07-3
- Epidanshenspiroketallactone
Catalog No.:BCN3142
CAS No.:113472-19-8
- Rhein-8-glucoside calcium salt
Catalog No.:BCN6349
CAS No.:113443-70-2
- VER 155008
Catalog No.:BCC2338
CAS No.:1134156-31-2
- KN-92 phosphate
Catalog No.:BCC1682
CAS No.:1135280-28-2
- Altanserin hydrochloride
Catalog No.:BCC7183
CAS No.:1135280-78-2
- 6-Bnz-cAMP sodium salt
Catalog No.:BCC8043
CAS No.:1135306-29-4
- 25(S)-Hydroxyprotopanaxatriol
Catalog No.:BCN2495
CAS No.:113539-03-0
- Ac-IEPD-AFC
Catalog No.:BCC2358
CAS No.:1135417-31-0
- Magnoloside A
Catalog No.:BCN6013
CAS No.:113557-95-2
- 1,2,3,19-Tetrahydroxy-12-ursen-28-oic acid
Catalog No.:BCN1615
CAS No.:113558-03-5
- Ikarisoside F
Catalog No.:BCN2284
CAS No.:113558-14-8
- Baohuoside I
Catalog No.:BCN5350
CAS No.:113558-15-9
- E-4031 dihydrochloride
Catalog No.:BCC7182
CAS No.:113559-13-0
- Q-VD-OPh hydrate
Catalog No.:BCC1125
CAS No.:1135695-98-5
- Tigecycline mesylate
Catalog No.:BCC4229
CAS No.:1135871-27-0
Entecavir maleate versus entecavir in Chinese chronic hepatitis B predominantly genotype B or C: Results at week 144.[Pubmed:28345157]
J Viral Hepat. 2017 Oct;24(10):877-884.
Reports on the efficacy and safety of long-term entecavir treatment in chronic hepatitis B (CHB) predominantly genotype B or C are insufficient. This study presents the efficacy and safety of entecavir maleate in Chinese CHB patients. Patients were randomly assigned to receive 48-week treatment with either 0.5 mg/day entecavir (group A) or 0.5 mg/day entecavir maleate (group B), and then all patients received treatment with 0.5 mg/day entecavir maleate from week 49. Two hundred and seventy-five patients with CHB (HBeAg-positive: 218) were analysed, predominantly (98.5%) with genotype B or C. Baseline characteristics were balanced. For the HBeAg-positive CHB patients, the mean HBV DNA level decreased similarly (A: by 6.36 log10 IU/mL vs B: by 6.31 log10 IU/mL) between groups at week 144. The percentages of patients who achieved undetectable HBV DNA were similar (A: 70.59% vs B: 66.67%) between groups. Similar HBeAg loss rates (A: 43.53% vs B: 40.23%; P>.05) and HBeAg seroconversion rates (A: 21.52% vs B: 21.18%) were achieved. For the HBeAg-negative CHB patients, similar reductions in HBV DNA levels from baseline (A: by 6.13 log10 IU/mL vs B: by 5.65 log10 IU/mL) and percentages of patients who achieved undetectable HBV DNA (A: 100% vs B: 100%) were achieved. The overall incidence of adverse events was comparable between groups. In conclusions, 48-week administration of entecavir maleate and entecavir showed similar efficacy and safety in Chinese patients with CHB. Long-term entecavir maleate treatment was effective and safe in CHB patients.
Determination of fluvoxamine maleate in human urine and human serum using alkaline KMnO4 -rhodamine B chemiluminescence.[Pubmed:28371383]
Luminescence. 2017 Sep;32(6):1077-1083.
The flow-injection chemiluminescence (FI-CL) behavior of a gold nanocluster (Au NC)-enhanced rhodamine B-KMnO4 system was studied under alkaline conditions for the first time. In the present study, the as-prepared bovine serum albumin-stabilized Au NCs showed excellent stability and reproducibility. The addition of trace levels of fluvoxamine maleate (Flu) led to an obvious decline in CL intensity in the rhodamine B-KMnO4 -Au NCs system, which could be used for quantitative detection of Flu. Under optimized conditions, the proposed CL system exhibited a favorable analytical performance for Flu determination in the range 2 to 100 mug ml(-1) . The detection limit for Flu measurement was 0.021 mug ml(-1) . Moreover, this newly developed system revealed outstanding selectivity for Flu detection when compared with a multitude of other species, such as the usual ions, uric acid and a section of hydroxy compounds. Additionally, CL spectra, UV-visible spectroscopes and fluorescence spectra were measured in order to determine the possible reaction mechanism. This approach could be used to detect Flu in human urine and human serum samples with the desired recoveries and could have promising application under physiological conditions.
[Chinese expert consensus on the use of topical timolol maleate treatment of infantile hemangiomas].[Pubmed:28275803]
Shanghai Kou Qiang Yi Xue. 2016 Dec;25(6):744-747.
Non-selective beta-blocker propranolol has been proved by FDA as the first-line agent for infantile hemangioma (IH) with dramatic response. To reduce the side effects caused by systemic administration of propranolol, timolol maleate treatment has been increasingly used as an alternative to systemic beta-blockers and watchful waiting for many IH patients in recent years. However, the appropriate indications, drug dosage, dosing regimen, time for initiation, optimal duration, monitoring for side effects still remains controversial. To standardize the use of topical timolol in treating IH, avoid overtreatment or under-treatment, as well as minimize complications, a Chinese expert consensus on the use of topical timolol treatment of IH has been approved and written by a multidisciplinary experts group based on an up-to-date literature review and repeated discussion, which can be used to reduce inappropriate variations in clinical practice and to promote the delivery of high quality, evidence-based health care for IH patients.
Evaluation of the effects on choroidal thickness of bimatoprost 0.03% versus a brinzolamide 1.0%/timolol maleate 0.5% fixed combination.[Pubmed:28376651]
Cutan Ocul Toxicol. 2017 Dec;36(4):397-403.
OBJECTIVE: To investigate the effects of two different medical treatment options on choroidal thickness (CT) in cases of open-angle glaucoma (OAG). METHODS: Sixty-seven eyes newly diagnosed with OAG and 52 healthy eyes constituting the control group were included in the study. Glaucomatous eyes were randomly divided into two subgroups; Group I was started on bimatoprost 0.03% and Group II on a brinzolamide 1.0%/timolol maleate 0.5% fixed combination (BTFC). Intraocular pressure (IOP), ocular pulse amplitude (OPA) and subfoveal CT measurements were performed in all eyes in the study before treatment and on weeks 2, 4 and 8 after treatment. RESULTS: Mean initial IOP values in groups I and II and the control group were 25.5 +/- 4.7, 25.1 +/- 5.2 and 16.1 +/- 2.9 mmHg, mean OPA values were 3.7 +/- 1, 3.6 +/- 1.4 and 2.4 +/- 0.6 mmHg and mean CT values were 269.4 +/- 83, 264.5 +/- 84.4 and 320.1 +/- 56.6 mum, respectively. Eight weeks after treatment, mean IOP values in Groups I and II and the control group were 18.3 +/- 2.6, 18.1 +/- 3.4 and 15.7 +/- 2.9 mmHg, mean OPA values were 2.9 +/- 1.2, 2.8 +/- 1.5 and 2.3 +/- 0.8 mmHg and mean CT values were 290.2 +/- 87.3, 271.8 +/- 82.5 and 319.3 +/- 56.8 mum, respectively. No significant difference was determined in terms of the decrease in IOP and OPA obtained after treatment in Group I and Group II. However, a significant difference was observed between the two groups in terms of choroidal thickening after treatment. CONCLUSION: The use of topical ocular hypotensive medication in eyes with OAG results in an increase in CT. This increase is relatively greater with bimatoprost 0.03% therapy compared to BTFC.
Effect of N-alkylation on the affinities of analogues of spiperone for dopamine D2 and serotonin 5-HT2 receptors.[Pubmed:1531364]
J Med Chem. 1992 Feb 7;35(3):423-30.
Two series of N-substituted spiperone analogues were prepared and evaluated in vitro to measure their affinities for dopamine D2 and serotonin 5-HT2 receptors. Substitution of the amide nitrogen with an alkyl group of five carbon units or less resulted in analogues displaying a low selectivity for D2 compared to 5-HT2 receptors. However, a moderate improvement in selectivity for D2 receptors was observed with N-benzylspiperone. Substitution at either the ortho or para position of the benzyl group resulted in a further reduction in affinity for 5-HT2 receptors and improvement in the selectivity ratio. Examination of N-substituted analogues of spiperone may provide insights into the topography of the antagonist binding region of the 5-HT2 receptor. The results also suggest that an 18F-labeled analogue of N-(4-nitrobenzyl)spiperone (4p) may be a suitable tracer for studying D2 receptors with positron emission tomography since this compound displays a high selectivity for D2 receptors relative to that of spiperone and N-methylspiperone.


